e. Setting Reactions. Unlike zinc phosphate cement, the setting reaction of
polycarboxylate cement produces little heat. This has made it a material of choice.
Manipulation is simpler and trauma due to thermal shock to the pulp is reduced. The
rate of setting is affected by the powder-liquid ratio, the reactivity of the zinc oxide, the
particle size, the presence of additives, and the molecular weight and concentration of
the polyacrylic acid. The strength can be increased by additives such as alumina and
fluoride. The zinc oxide reacts with the polyacrylic acid forming a cross-linked structure
of zinc polyacrylate. The set cement consists of residual zinc oxide bonded together by
a gel-like matrix.
1-16. PREPARATION AND USAGE OF POLYCARBOXYLATE CEMENT
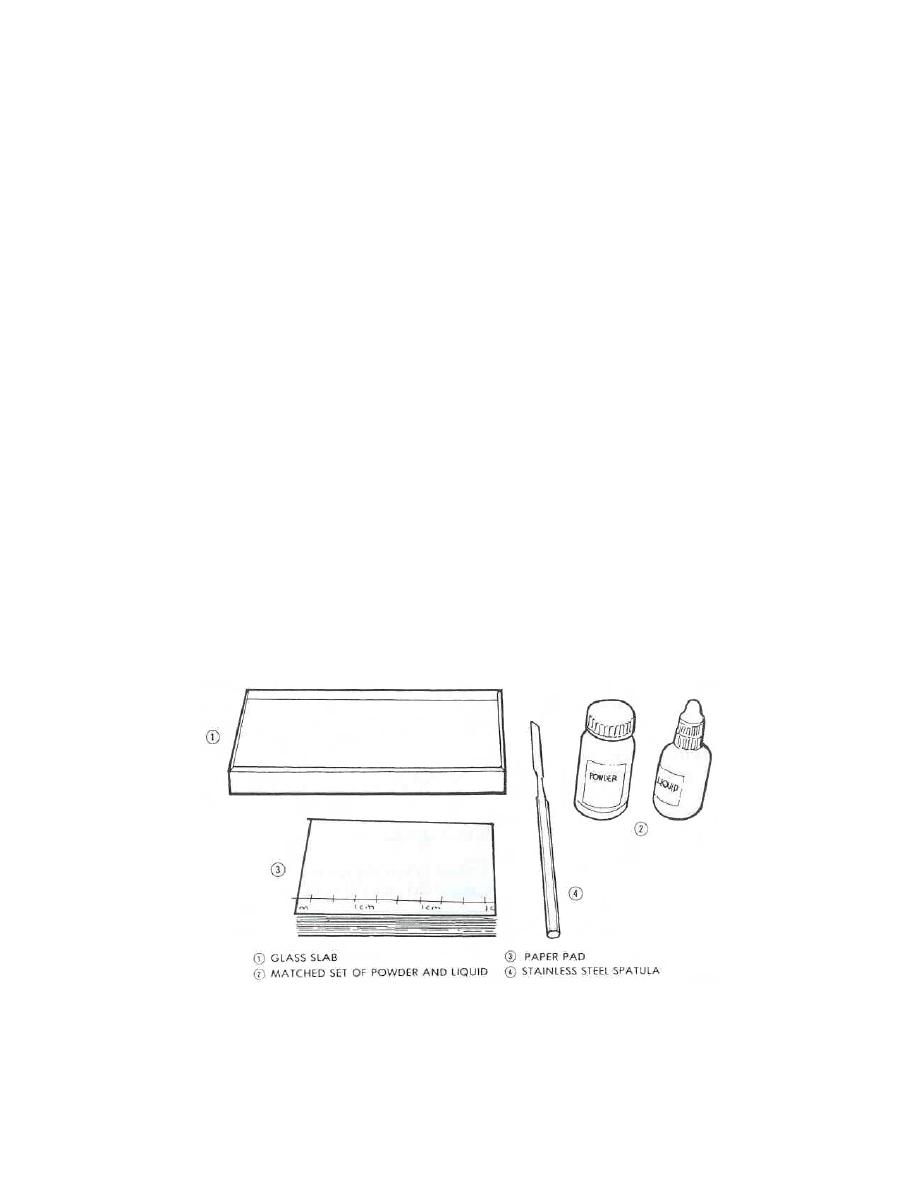
a. Equipment. The equipment required for mixing polycarboxylate cement
consists of a nonporous, polymer paper pad, a glass mixing slab, a stainless steel
spatula, and a matched set of powder and liquid (see figure 1-5).
b. Powder and Liquid. Dispense the powder and liquid according to
manufacturer's instructions (to achieve the desired consistency). Do not predispense
and allow to sit. Loss of moisture will cause the liquid to thicken.
c. Mixing. Mixing is done by rapidly blending the powder and the liquid for 30
seconds on a polymer paper-mixing pad. Ensure that all the powder is incorporated into
the mix. If extended working time is desired, mix on a cooled glass slab.
d. Characteristics of a Completed Mix. The correct cementing mix is more
viscous than zinc phosphate cement. Because of its composition, the cementing mix
flows adequately under pressure.
Figure 1-5. Setup for mixing polycarboxylate cement.
MD0502
1-16



 Previous Page
Previous Page
