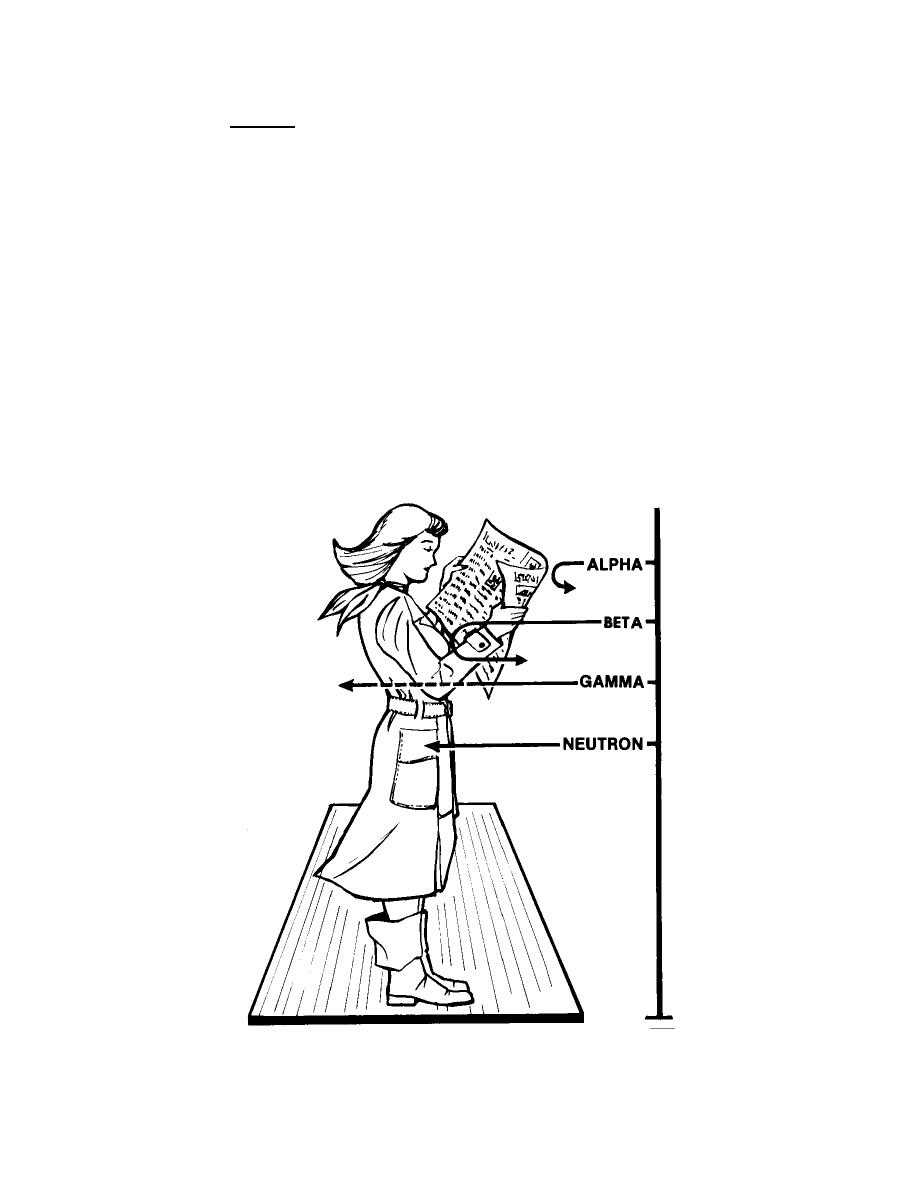
(4) Neutron. A neutron is an electrically neutral or uncharged particle of
matter which exists along with protons in the nucleus of all elements except the
mass/isotopy of hydrogen.
c. At one time, scientists believed that the atom was the basic particle of all
matter. Further inquiry and research revealed that the atom is actually composed of
several smaller particles: electrons, protons, and neutrons. The proton has a positive
charge, the electron has a negative charge, and the neutron has no electrical charge.
In a specific element, all the atoms have the same number of electrons and protons.
For example, all sodium atoms have the same number of electrons and protons, and all
calcium atoms have the same number of electrons and protons. A sodium atom,
however, and a calcium atom do not have the same number of electrons and protons.
Until an atom's nuclear structure is excited by the addition of energy or mass, an atom is
physically stable and electrically neutral. When the nucleus of an atom becomes
excited, it becomes unstable and will of its own accord revert to its original stable
condition. Subatomic particles (gamma radiation) are emitted during this change. This
change during which there is a nuclear emission of mass or energy is called
radioactivity, and the products released by radioactivity are called nuclear radiation.
Figure 2-1. Four basic types of nuclear radiation.
MD0587
2-3



 Previous Page
Previous Page
