3-3.
STRUCTURAL FORMULAS
A structural formula is a chemical formula that shows how atoms are bonded to
each other. For example, we might write AlOHCl2 as
Cl1
OH
Al
Cl1
to show the bonds. However, in inorganic chemistry, the compounds are such that
there is generally only one possible way to combine the atoms. This is not the case in
organic chemistry, where very often there are many possible combinations for the
atoms in the compound. Consider, for example, the formula C4H10. This formula could
represent either of the following compounds.
CH3-CH-CH3
or
CH3-CH2-CH2-CH3
CH3
These compounds have slightly different properties. As the formulas become more
complex, the differences are even greater. For this reason, it is often better to use a
structural formula in organic chemistry rather than the simple chemical formula.
3-4.
CARBON
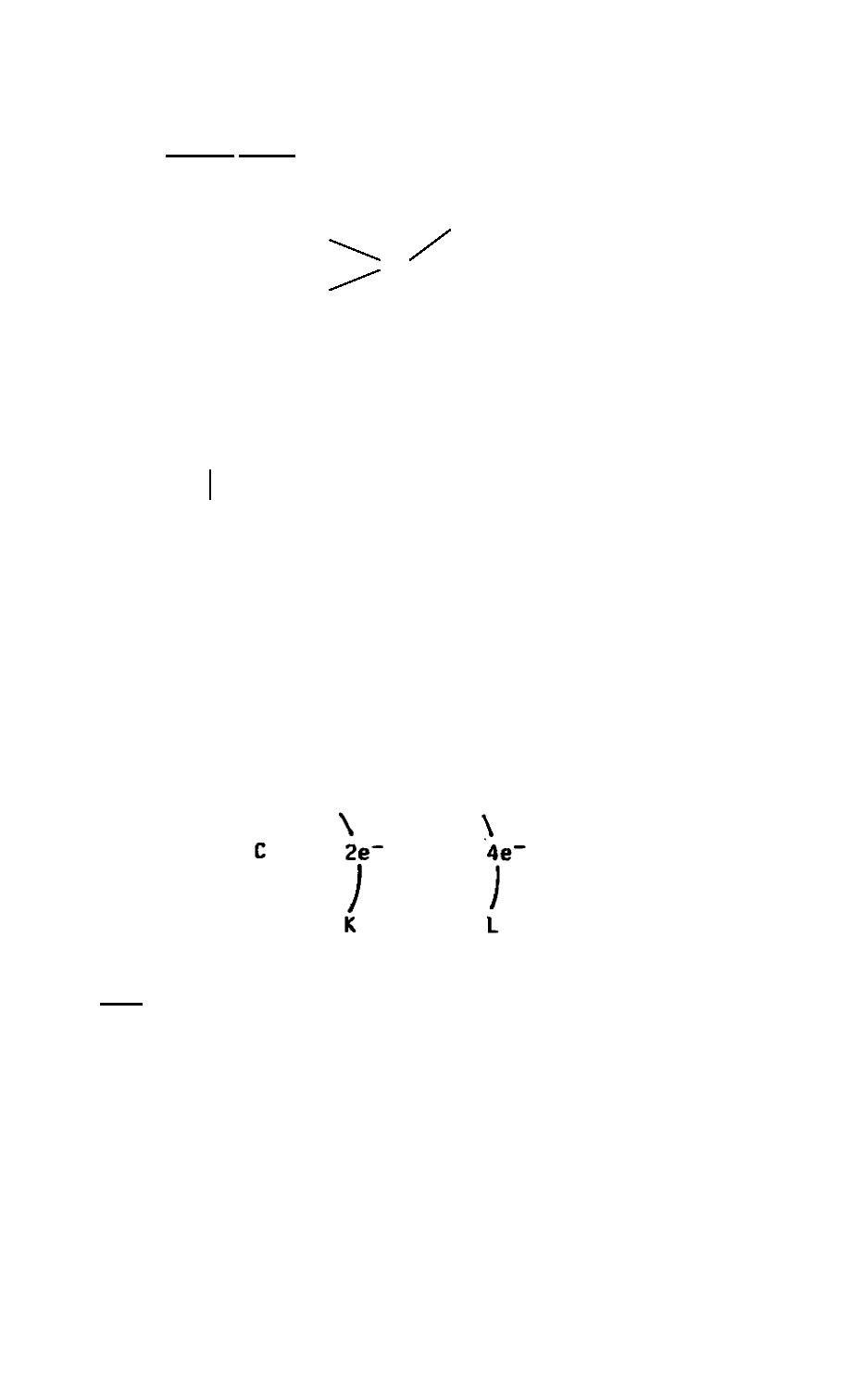
Before we examine carbon compounds, we first need to examine the structure
and mention some properties of the carbon atom. Carbon has an atomic number of six,
meaning it has six protons, and consequently has six electrons. These electrons are
distributed with two in the K shell and four in the L shell. In forming compounds, carbon
would appear to gain or lose the four electrons in its outer shell. Thus, we have the +4,
-4 valences you learned for carbon earlier in this subcourse.
In fact, carbon does not usually exchange electrons with other elements but prefers to
share four electrons to complete its L shell. This is the reason that covalent bonding is
predominant in organic chemistry.
3-3
MD0803




 Previous Page
Previous Page
