(1) In fact, the acidity of a solution is the concentration of hydrogen ions in
that solution. Since we can calculate the hydrogen ion concentration, as you will learn
later, we can now determine a numerical value of the acidity of a solution. The
concentrations of hydrogen ions in both acidic and basic solutions are generally very
small. A strong solution of HCl, for example, may contain only 0.01 mole of hydrogen
ions per liter of solution. A solution of NaOH may have as little as 0.00000000001 mole
of hydrogen ion per liter of solution.
(2) To simplify the expression of such terms, chemists have transformed the
concentration values into numbers, called pH numbers, which are easier to utilize. This
is done according to the following equation:
pH = -log[H+]
The abbreviation log stands for logarithm. (For example, log 1 = 0, log 0.1 = -1, log 0.01
hydrogen ions in moles per liter. If we consider the two previous examples, you can see
how this transformation aids us. The pH of the HCl solution would be -(-2.0) = 2.0; the
pH of the NaOH solution would be -(-11.0) = 11.0. These numbers, 2 and 11, are
certainly easier to work with than 0.01 and 0.00000000001.
b. pH Scale. This transformation results in a range of pH numbers from 0 to 14,
which is called the pH scale.
(1) The limits of the scale are related to the dissociation; how they are
arrived at is beyond our scope. Further information on this relationship can be found in
an inorganic chemistry textbook.
(2) While you will not need to calculate a pH value, you will need to interpret
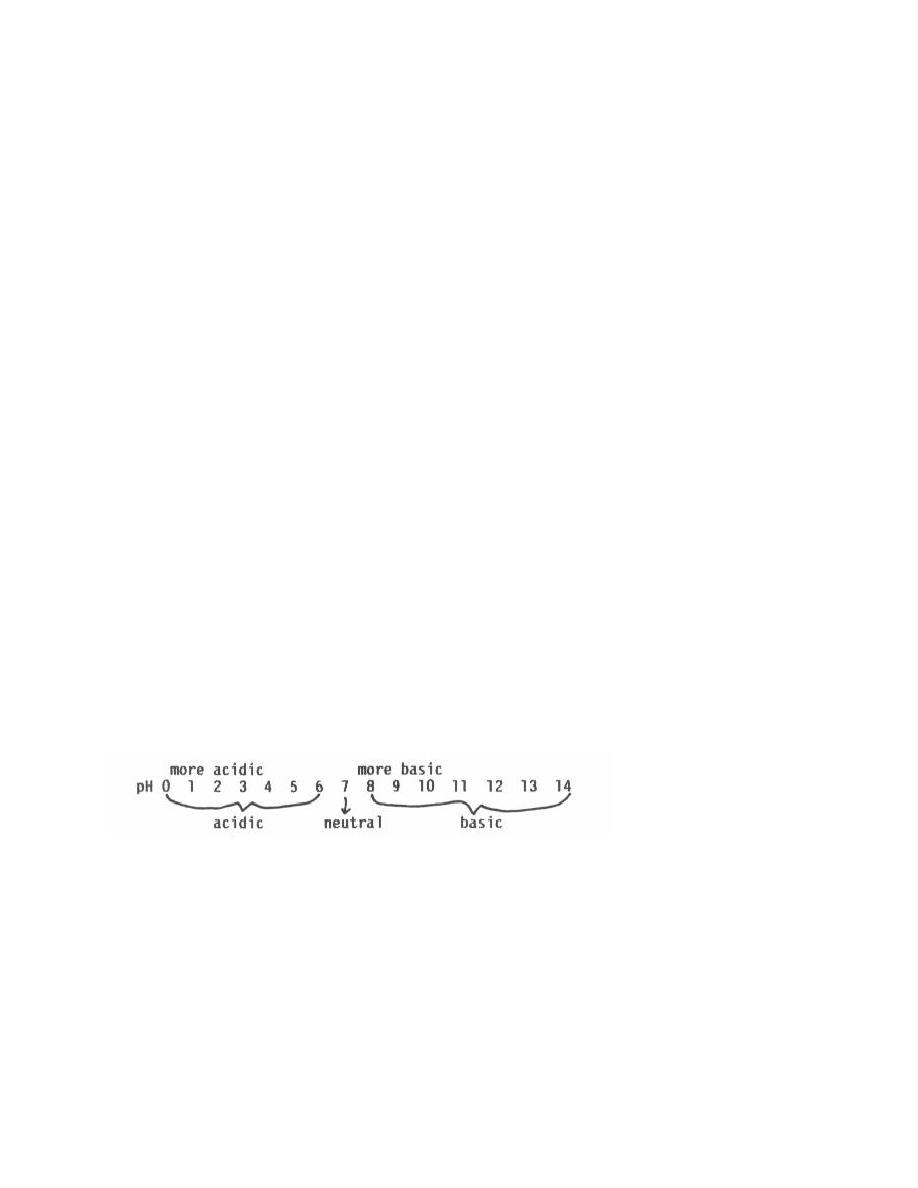
what a pH value means at times. To learn this function, examine the following pH scale:
(3) A pH value less than 7.0 means the solution is acidic; the lower the
number, the more acidic. A solution with a pH of 2.0 is more acidic than one with a pH
of 4.0. Any pH value greater than 7.0 means the solution is basic with larger numbers
indicating solutions that are more basic. The only value on the scale that indicates a
neutral solution is 7.0. The pH values for some common pharmaceutical products are
given below.
MD0803
2-20



 Previous Page
Previous Page
