5-4.
IMMUNODIFFUSION
Immunodiffusion techniques detect antigen-antibody precipitation reactions in a
semisolid medium. The formation of antigen and antibody complexes can be influenced
by a number of factors: relative concentration of antigens and antibodies, ionic strength
of the buffer, pH, and temperature. Two techniques most often used in a clinical
laboratory are single and double diffusion.
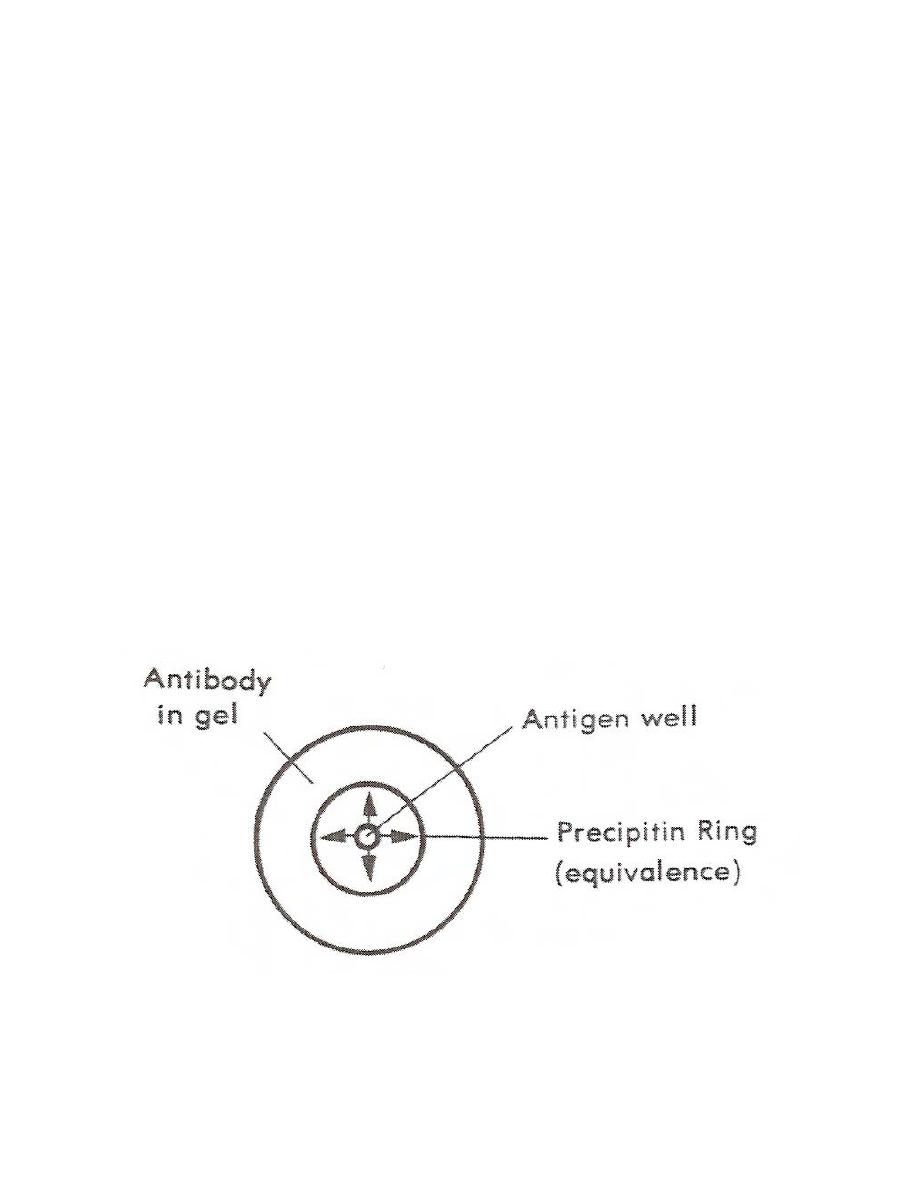
a. Radial Immunodiffusion (RID). In this method, a known concentration of
antibody is incorporated into an agarose medium. The reactant (antigen) is applied to a
well cut in the agarose and radially diffuses from the site of application (Figure 5-1). At
the point of equivalence, the antigen and antibody react to form a visible precipitin ring.
The size of the precipitin ring is proportional to the concentration of the antigen. In the
clinical laboratory, radial immunodiffusion is primarily used to quantitate serum
immunoglobulins and complement components such as C3 or C4.
b. Double Diffusion (DD). DD is based on the principle that antigen and
antibody diffuse through a semisolid medium and form a precipitin line. In Ouchterlony's
method, a layer of agar gel is deposited in a petri dish and circular wells are punched
out near one another in the gel. Antibody is then added to one well while antigen is
added to the other. These materials are allowed to diffuse radially from their respective
wells. As the perimeter of the diffusing substance increases, the concentration of that
substance within the perimeter continually decreases. When the optimal concentration
of antigen and antibody is reached, a line of precipitation is formed in the gel. The
precipitin line is relatively straight and is perpendicular to the axis line between the two
wells. The immunologic reactions in double gel diffusion are of three types (Figure 5-2):
Figure 5-1. Single immunodiffusion.
MD0838
5-3



 Previous Page
Previous Page
