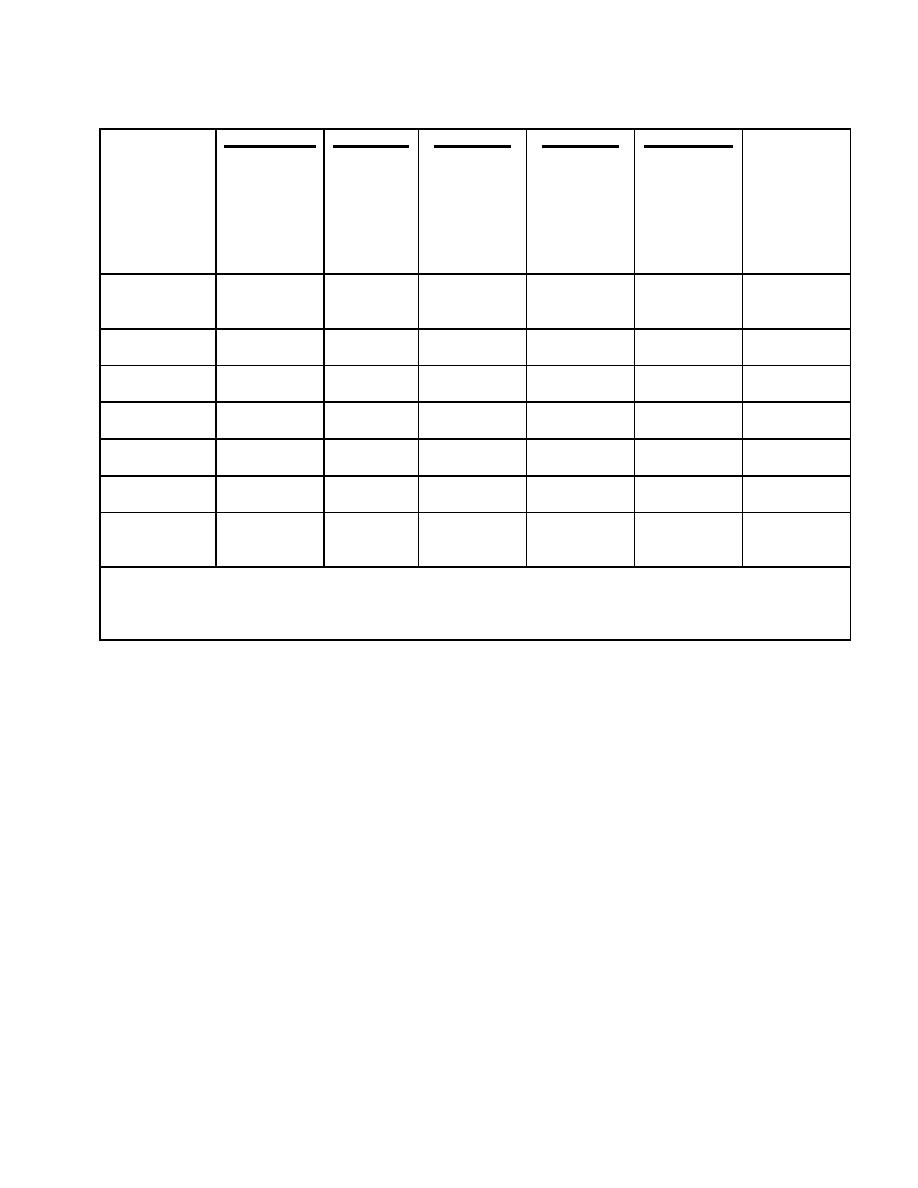
____CPD___
_________
_________
_________
__CPDA-1__
_________
Whole
Whole
Whole
Red
Whole
Red
Blood
Blood
Blood
Blood
Blood
Blood
Cells
Cells
Variable Days
of Storage
0
21
0
0
35
35
% viable cells
100.0
80.0
100.0
100.0
79.0
71.0
(24hr
posttranfusion)
7.2
6.84
7.6
7.55
6.98
6.71
pH (measured
at 37C)◦
ATP (% initial
100.0
86.0
100.0
100.0
56.0 (+/-16)
45.0 (+/-12)
value)
2.3-DPG (% of
100.0
44.0
100.0
100.0
<10.0
<10.0
initial value)
Plasma K+
3.9
21.0
4.2
5.1
27.3
78.5*
(mEq/1)
Plasma Na+
168.0
156.0
169.0
169.0
155.0
111.0
(mEq/1)
Plasma
1.7
19.1
8.2
7.8
46.1
658*
hemoglobin
(mg/dl)
* Values for plasma hemoglobin and potassium concentrations may appear somewhat high in 35-day stored
red blood cell units; the total plasma in these units is only about 70 ml.
Table 1-2. Biochemical changes of blood stored in CPD and CPDA-1.
(10) Stored red cells, even those at the end of their allowable shelf-life, can
be rejuvenated with FDA-approved solutions containing pyruvate, inosine, phosphate,
adenine, and in some cases, glucose. The rejuvenated red cells have increased levels
of 2,3-DPG and ATP. Rejuvenated red cells can be frozen and stored in the same
manner as fresh red cells and demonstrate normal oxygen transport when transfused.
b. Heparin. Heparin exerts its anticoagulant effect by potentiating the action of
the endogenous plasma protein, antithrombin III (AT III). Synthesized in the liver, AT III
is an inhibitor of most serine esterase clotting factors. Because it lacks dextrose,
heparin serves only as an anticoagulant, not as a preservative. Heparinized blood must
be transfused within 48 hours, preferably within 24 hours. Heparin is not recommended
for routine blood collection.
MD0845
1-38



 Previous Page
Previous Page
