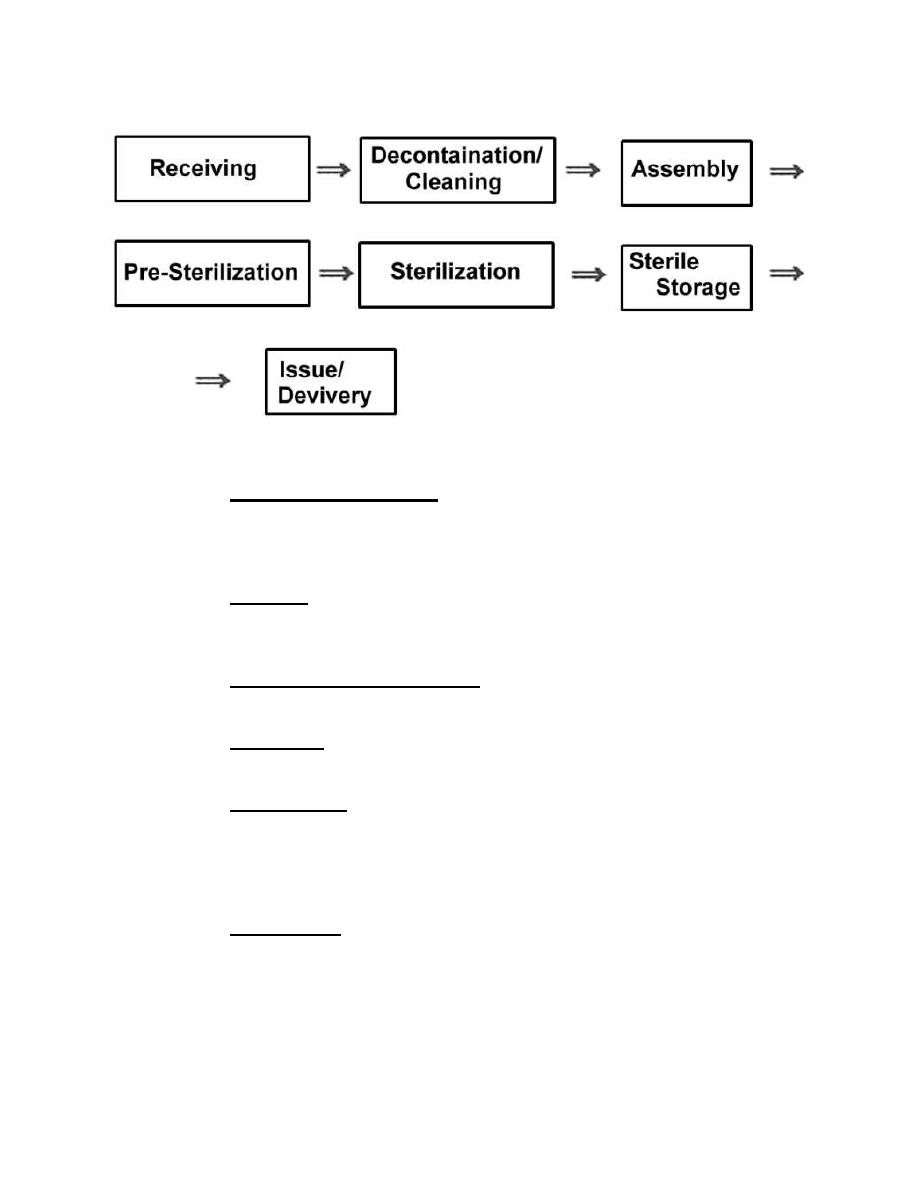
Figure 2-1. Workflow diagram - sequence of processing through the CMS
(2) Decontamination/cleaning. Items received by CMS are further cleaned
to the point they are safe for the CMS staff to handle and are ready to be assembled
into instrument sets, packs, trays, and so forth. This step may include terminal
sterilization.
(3) Assembly. Utilizing the Kardex, clean instruments and utensils,
equipment is arranged into the appropriate sets, packs, trays, and wrapped for
sterilization.
(4) Pre-sterilization storage (clean). Items awaiting the actual process of
sterilization are shelved or placed on carts until they are placed in the sterilizers.
(5) Sterilization. Assembled trays, sets, and packs are loaded into the
appropriate sterilizer (steam or gas) and undergo sterilization.
(6) Sterile storage. Following a sterilization cycle, items are removed from a
sterilizer marked with load number and expiration date, and placed on shelves until
such time as they are issued for use by CMS customers such as wards, clinics, and
TMCs. Items that have been purchased from the manufacturer in the sterile/ready-to-
use state are also shelved in the sterile storage area until they are issued.
(7) Issue/delivery. This is the point at which CMS items exit CMS--to be
transported to the patient care areas where they will be utilized.
MD0937
2-4



 Previous Page
Previous Page
