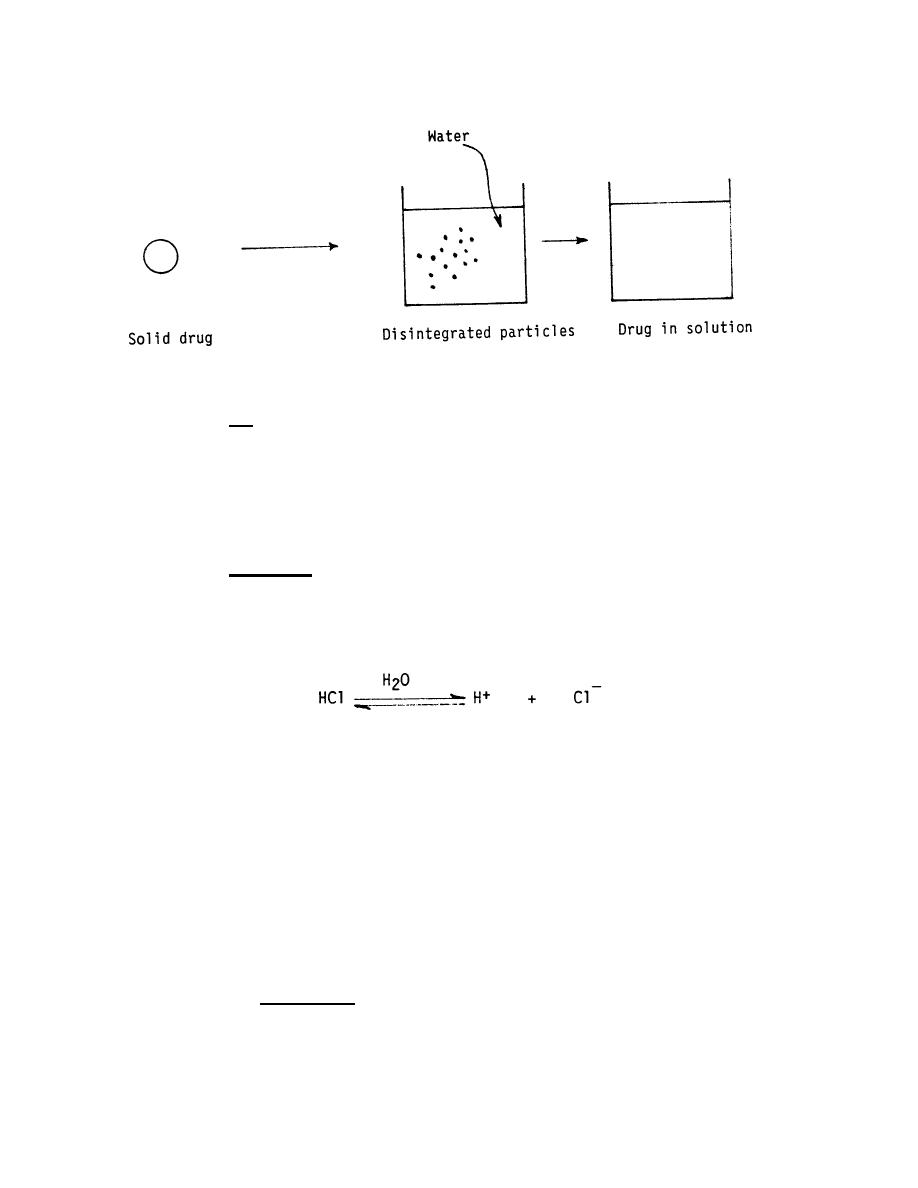
Figure 3-1. Dissolution of a drug.
(2) pH. The relative acidity or basicity of the fluids into which a drug is
placed will affect how rapidly the drug will dissolve. The pH of the stomach can be as
low as 1.0 (very acidic), the pH of the small intestine can range from 6.9 to 7.4 (slightly
acidic to slightly basic), while the pH of the plasma is approximately 7 4 (slightly basic).
Weakly acidic drugs (that is, aspirin) are more soluble in a basic or alkaline solution like
the small intestine (pH above 7.4). Weakly basic drugs, such as tetracycline
hydrochloride, are more soluble in an acidic solution like the stomach (pH below 7.0).
(3) Ionization. Ionization is the process whereby a substance breaks down
into positively and negatively charged particles (Figure 3-2).
(a) For example, when hydrochloric acid ionizes, it forms hydrogen
ions (H+) and chloride ions (Cl-).
Figure 3-2.
The ionization of hydrochloric acid.
(b) Equilibrium is established based on the chemical nature of each
drug. That is, a certain percentage of the drug ionizes, while the rest remains as the
compound. In summary, dosage forms must go into solution. A solid dosage form must
disintegrate and dissolve before it can be absorbed. A suspension dosage form has
already been partially dissolved; the drug particles must dissolve before absorption can
occur. A solution dosage form contains a drug that has already been dissolved; thus,
no disintegration or dissolution is required before absorption can occur.
b. Fat Solubility. In the last area, the topic of water solubility was discussed.
For instance, a drug is in solution. What other factors must it overcome in order to be
absorbed? One is fat solubility. Almost without exception, all body membranes are lipid
(fatty) in nature. Membranes separate even the various water compartments of the
body. These membranes are selectively permeable. That is, these membranes will
only allow certain materials to pass through them. In particular, the membranes favor
MD0804
3-13



 Previous Page
Previous Page
