b. Collisional Interactions. The discussion to this point has demonstrated how
x-rays are generated when electrons interact with the nucleus of an atom. X-rays are
also generated when electrons interact with the tightly bound orbital electrons of a hard
surface. This is called collisional interaction of an electron with matter and produces
characteristic or line radiation.
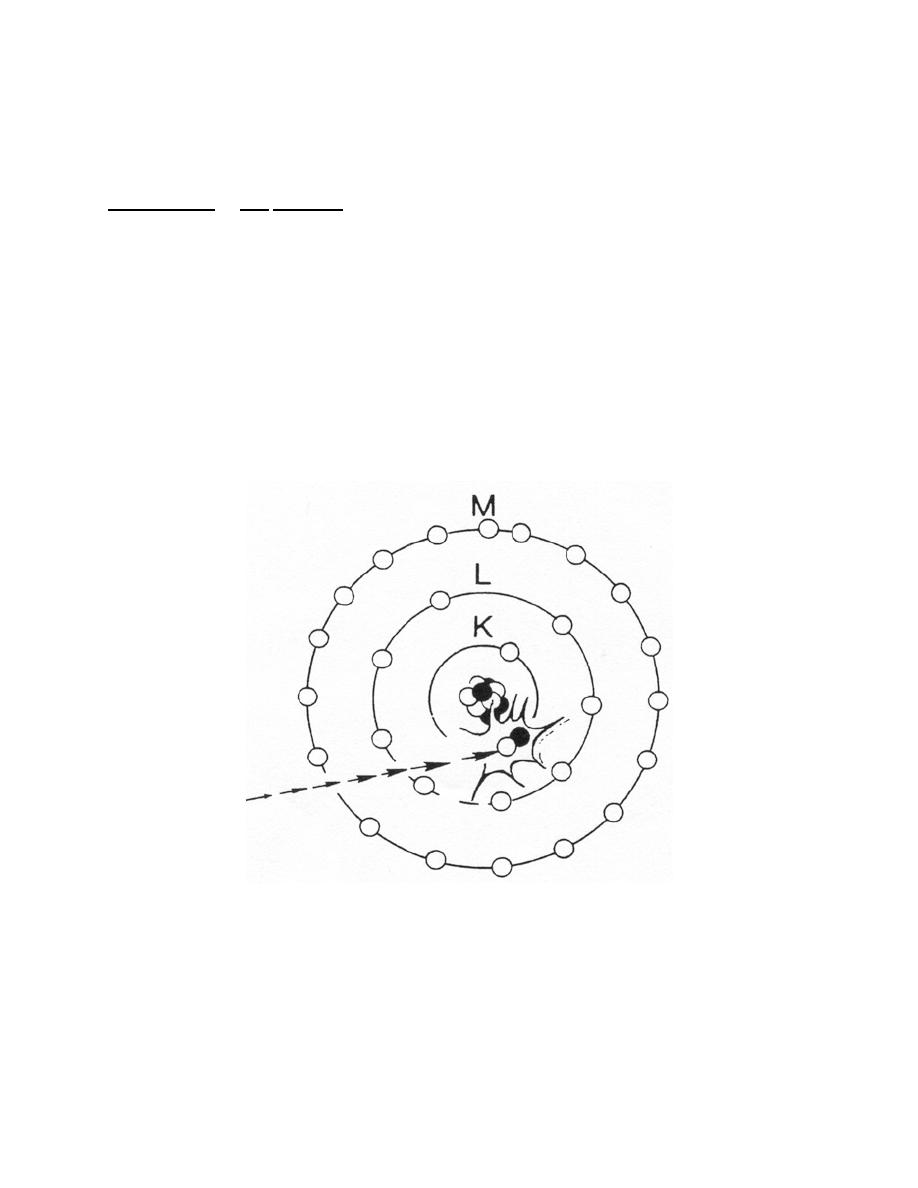
(1) In the interaction, an approaching electron collides with a tightly bound
orbital electron, such as an electron in the K shell of an atom of tungsten (figure 1-15).
Because of the collision, the K electron is ejected from its shell and energy is absorbed
by the atom equal to the binding energy of the shell. The atom is left in an excited state
or with an excess of energy and an electron vacancy in a shell. Immediately after
excitation, the atom returns to a normal state by emitting the energy it has absorbed in
the form of x-ray photons. It does this as follows. Another electron, such as from the L
shell, fills the vacancy. Since the potential energy of an electron in the L shell is higher
than that of an electron in the K shell, the L electron loses energy in the transition. The
energy lost is equal to the difference in the binding energies of the K and L shells and is
given off as an x-ray photon.
Figure 1-15. Collisional interaction in tungsten.
(2) Although a photon has been emitted, the process is not yet completed
because there is now a vacancy in the L shell and the atom still has an excess of
energy. This vacancy is also immediately filled by another electron such as one from
the H shell and another photon is emitted equal to the energy of the difference of the
transition. This chain reaction continues with a photon given off for each electron
transition until the atom returns to a normal state.
MD0950
1-20



 Previous Page
Previous Page
