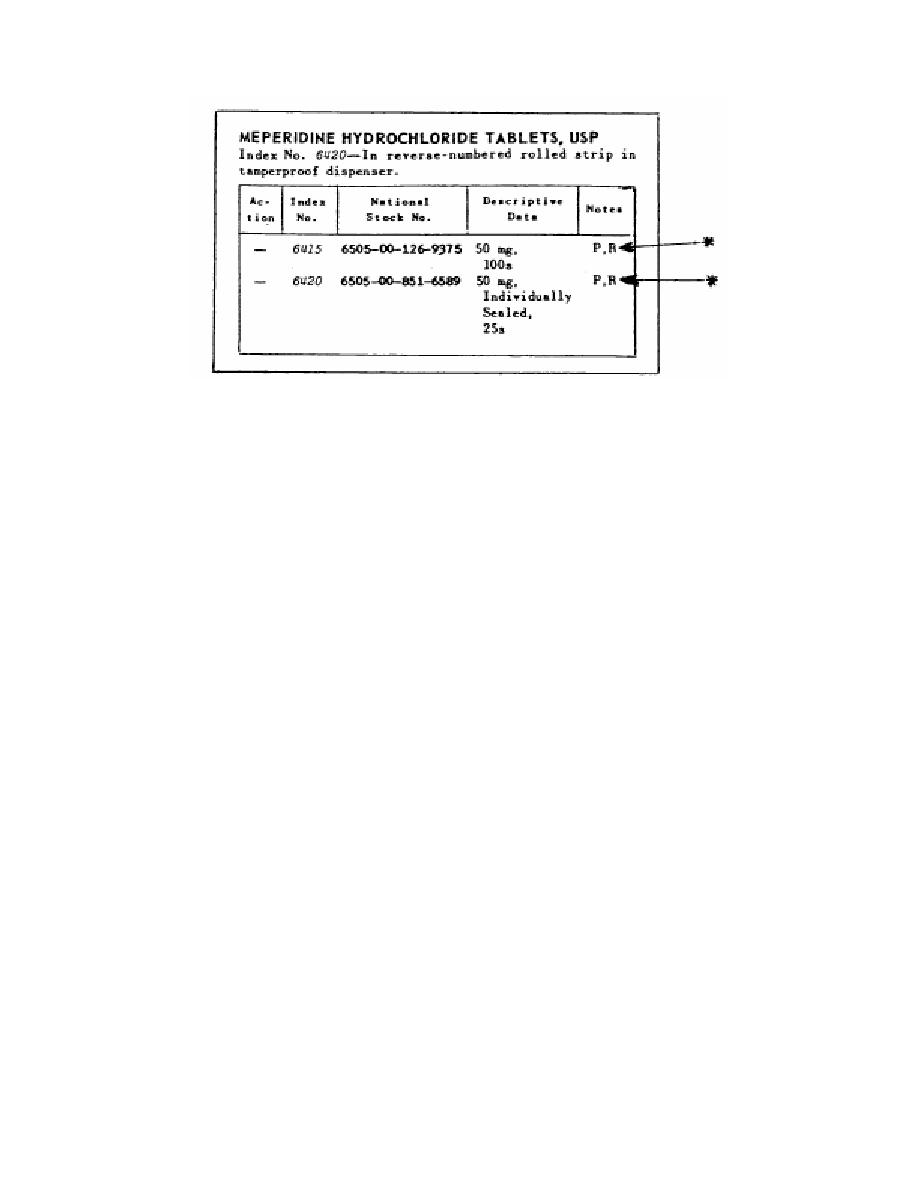
Figure 3-6. Note R Substance in Federal Supply Catalog.
3-5.
ACCOUNTABILITY PROCEDURES FOR NOTE Q AND NOTE R
SUBSTANCES
a. Introduction. Both Note Q and Note R substances must be rigidly controlled.
Prescriptions for these substances should be filled after the authority for the prescriber
to write for these medications has been verified. Further, records must be maintained to
show to whom, when, the quantity of the medication, and by whom the medication was
prescribed. Periodic inspections and inventories by disinterested personnel help to
ensure that the control substances are secured and dispensed following laws and
regulations.
b. Use of the Controlled Substances Stock Record (DA Form 3862). A DA
Form 3862 (figure 3-7) is maintained in the pharmacy to permanently record receipts
and expenditures of all controlled substances. A separate record must be maintained
on DA Form 3862 for each dosage form of a controlled substance maintained in the
pharmacy.
(1) Schedule II controlled substances, ethyl alcohol, and alcoholic liquors.
When an item is received, the date, the quantity received, and the voucher number will
be entered in the appropriate columns. Similarly, when stock is withdrawn, the date
expended, the prescription number or order number, and the amount expended will be
entered on the form. At the time the entry is made on the card--whether it be a receipt
or an expenditure--the amount involved will be added to or subtracted from, as
appropriate, the amount shown in the "Balance on Hand" column. The new figure will
always reflect the actual amount on hand.
(2) Schedule III, IV, and V controlled substances. When an item is
received, the date, the quantity received, and the voucher number will be entered in the
appropriate columns. Expenditures for each item will be summarized weekly and
posted to the DA Form 3862.
MD0810
3-10



 Previous Page
Previous Page
