LESSON 5
CONVERSION OF CONCENTRATION UNITS
5-1.
DISCUSSION
At times, units of concentration will be given, but another unit of concentration will
be required to perform calculations. In this lesson, we will discuss methods for
conversion from one unit of concentration to another.
5-2.
CONVERSION FROM mol/L TO g/dL AND FROM g/dL TO mol/L
Before discussing the methods for converting the above units, let's review the
definition for each unit.
EQUIVALENT WAYS OF EXPRESSING
CONCENTRATION (NO NUMERICAL CONVERSION NECESSARY)
g/dL = parts of solute per 100 parts
g/L = parts of solute per 1000 parts
molarity = moles per liter
The g/dL concentration is defined as parts of solute per 100 parts total solution.
The morlarity of a solution is defined as the number of moles per liter of solution (mol/L).
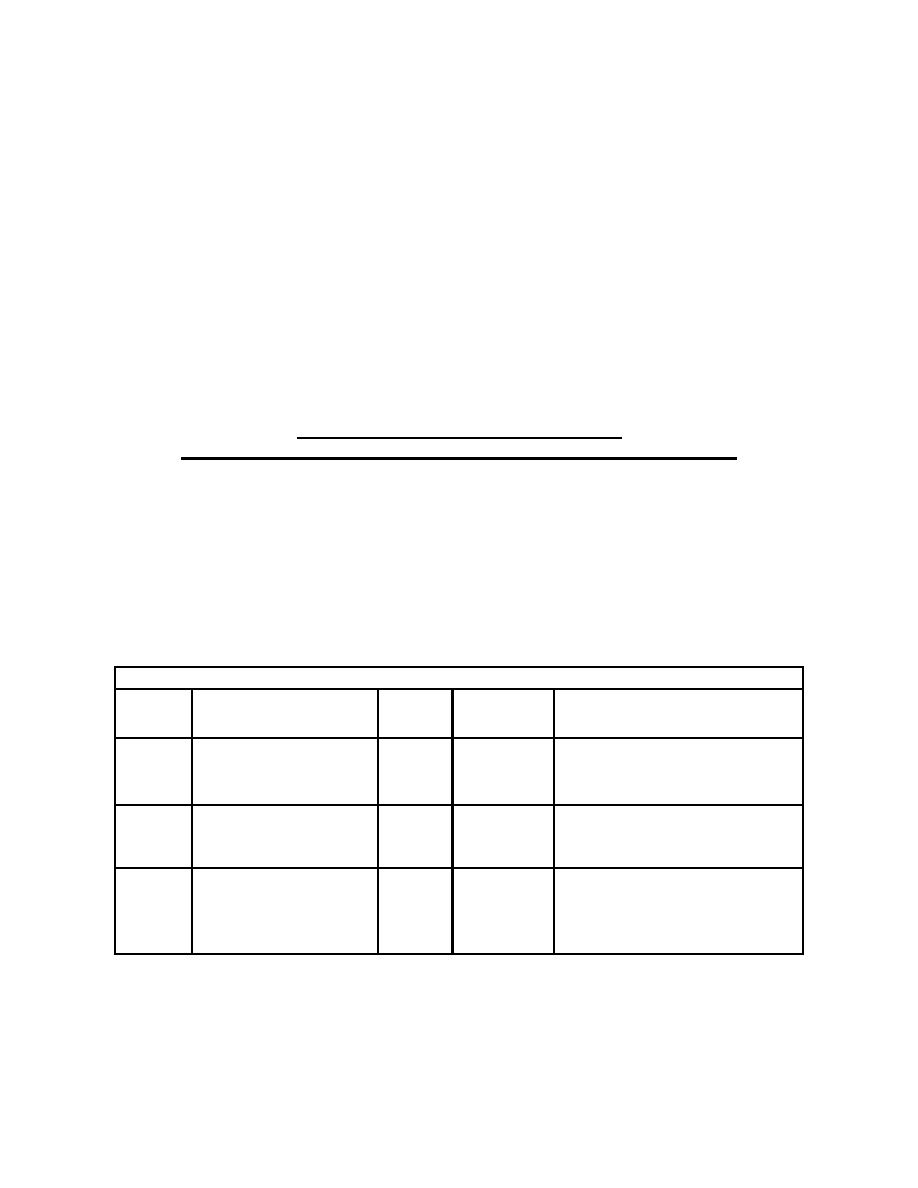
CONVERSIONS
CONCENTRATION
IN
TO GET CONCENTRATION
BY
EXPRESSED IN
UNITS
EXPRESSED IN
g/L or parts solute
Multiply
mol/L
GMW
g/mol
per 1000 parts
g/L or parts solute
Multiply
g/dL
10
d/L/L
per 1000 parts
dL mol
10
.
mol/L
Multiply
g/dL
------
-- ----
GMW
L
g
MD0837
5-2


 Previous Page
Previous Page
