b. Calculation. The following sample problems will illustrate the derivation
and use of the formula for mixing liquid concentrates with liquid diluents or solid
(powder) concentrates with solid (powder) diluents:
(1) Problem #1. 55 gallons of 1% diazinon emulsion are to be prepared
using diazinon, 48% emulsifiable concentrate and water. How many gallons of the
concentrate are required?
(a)
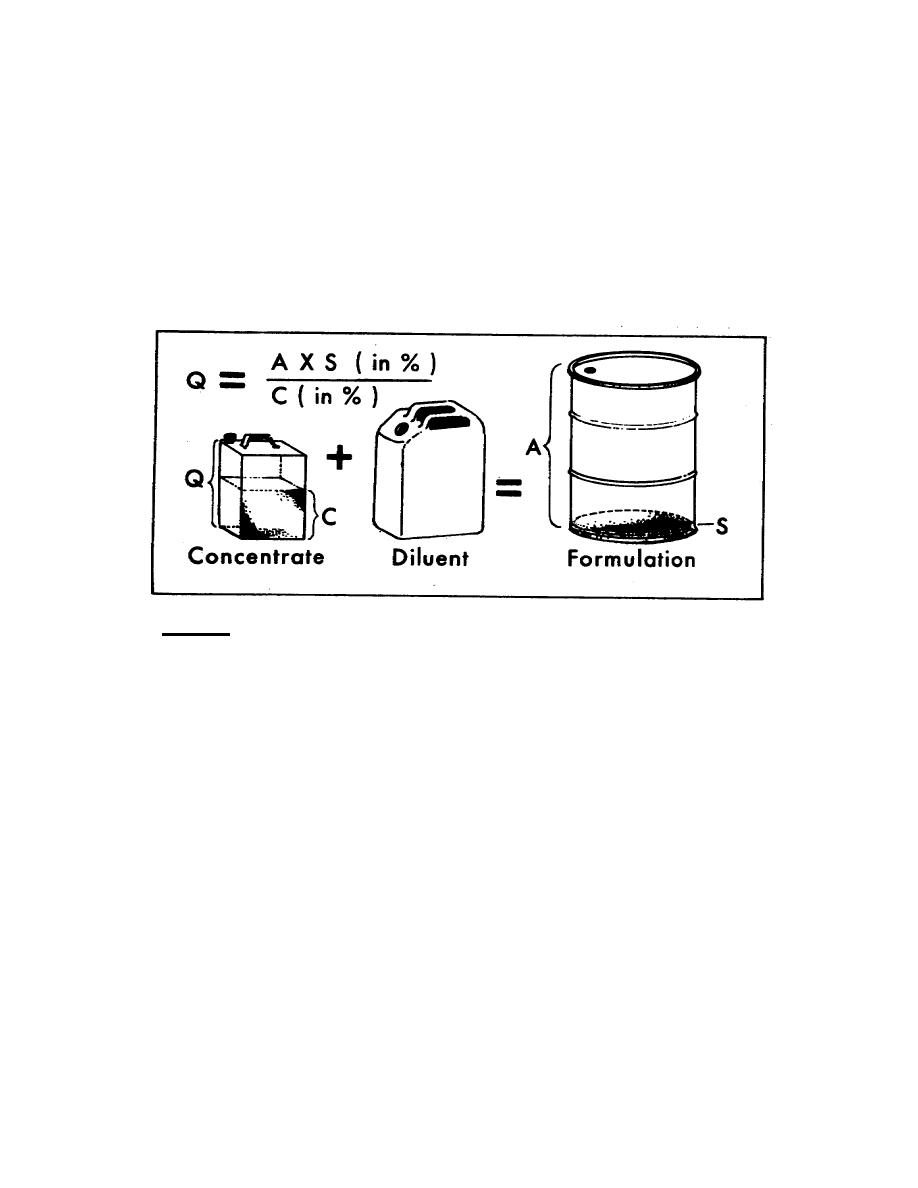
Figure 2-3 illustrates the problem graphically.
Notation:
Q = Quantity of concentrate in gallons or pounds (must be same units as A).
C = Concentration of toxicant in the concentrate in percent.
A = Amount of final formulation desired in gallons or pounds (must be same
units as Q).
S = Strength of final formulation in percent of active ingredient.
Figure 2-3. Volumetric calculations.
(b) Computation. The one element that will remain constant
throughout the problem is the actual amount of toxicant involved. When we dilute a
pesticide, we do not change the amount of toxicant. We merely add enough diluent so
that the final formulation will consist of the desired percentage of toxicant. The amount
of toxicant in either the concentrate or the final solution is determined by multiplying the
amount of concentrated or formulation by the percentage of toxicant. Thus:
MD0173
2-8



 Previous Page
Previous Page
