NOTE:
The electrons are shown orbiting the nucleus like planets orbiting a sun for
ease of explanation. The actual workings of the atom are a bit more
complicated.
Figure 2-3. Orbiting electrons: 10 electrons.
2-5.
ATOMS AND MOLECULES
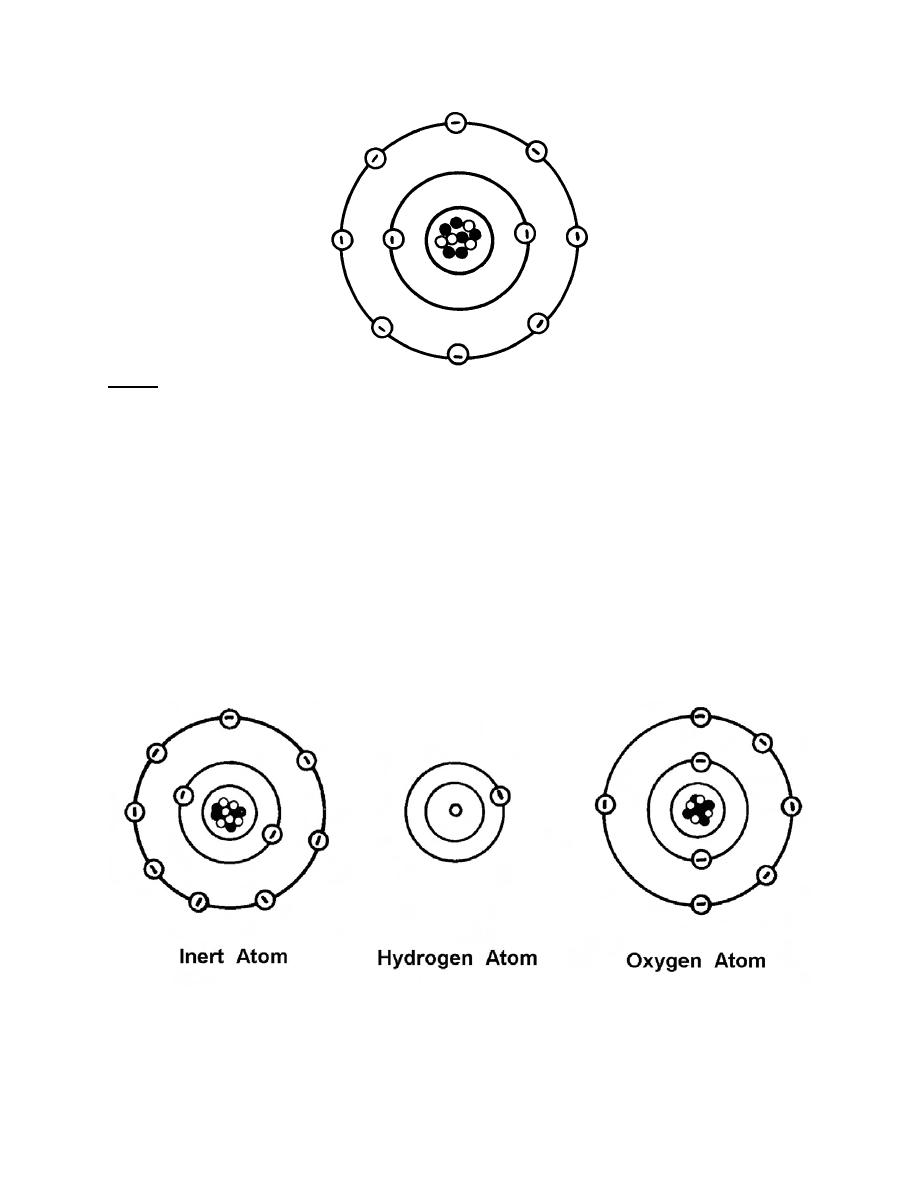
a. Atoms. Some atoms are inert or neutral atoms. They have outer shells that
are completely filled with electrons. (Remember, except for the K shell, an outer shell is
filled if it contains eight electrons.) It is extremely difficult for these atoms to combine
with other atoms to form molecules. Other atoms are referred to as bonding atoms.
They have less than eight electrons in their outer shell (or less than two electrons if the
K shell is the outer shell) and combine readily with atoms of other bonding atoms. See
figure 2-4.
Figure 2-4. Inert atoms and bonding atoms.
MD0512
2-4



 Previous Page
Previous Page
