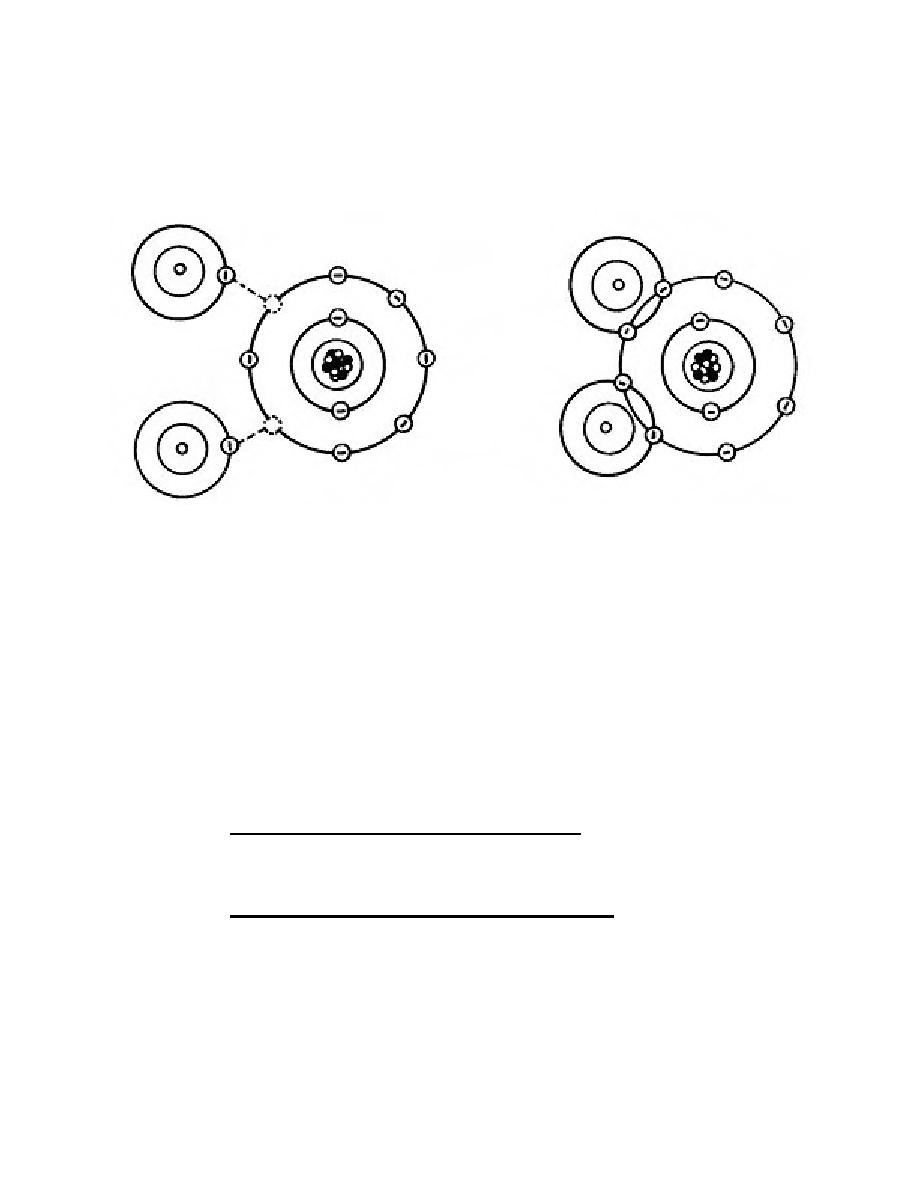
b. Molecules. Molecules are formed by the combination of two or more atoms.
A very common molecular formation is water, resulting from the combination of bonding
atoms. Both have outer shells containing less than eight electrons. Two hydrogen
atoms combine (or bond) with one oxygen atom to form water or H20. See figure 2-5.
2-6.
X-RAY PHOTON
A brief definition of an x-ray photon was previously stated. However, a more
comprehensive definition is necessary for the next series of photon actions. X-ray
photons are electromagnetic rays produced in the x-ray tube head when electrons from
the cathode filament strike the anode target. They are bundles of pure energy. The
photons transfer their energy to the substance through which they pass whether it be
air, an x-ray film, or the living tissue of the patient or the specialist. They cannot be
seen or felt.
a. Photon Action Upon Atoms.
(1) Photon collision with the nucleus of an atom. The photon may strike the
nucleus of an atom (figure 2-6). If this occurs, the atom will be destroyed and the
photon will release or expend its energy.
(2) A direct photon hit upon an electron by a photon. The photon may strike
an electron with a direct hit (figure 2-7). This action will result in the release of the
photon's energy, transferring its energy to the electron. The electron will be dislodged
from its shell. When an electron is dislodged in this manner, it is called a photo-
electron. The dislodged or departing electron (now a photo-electron) will have energy to
ionize or strike other electrons. This is a form of scattered/secondary radiation, as
noted earlier in the text.
MD0512
2-5



 Previous Page
Previous Page
