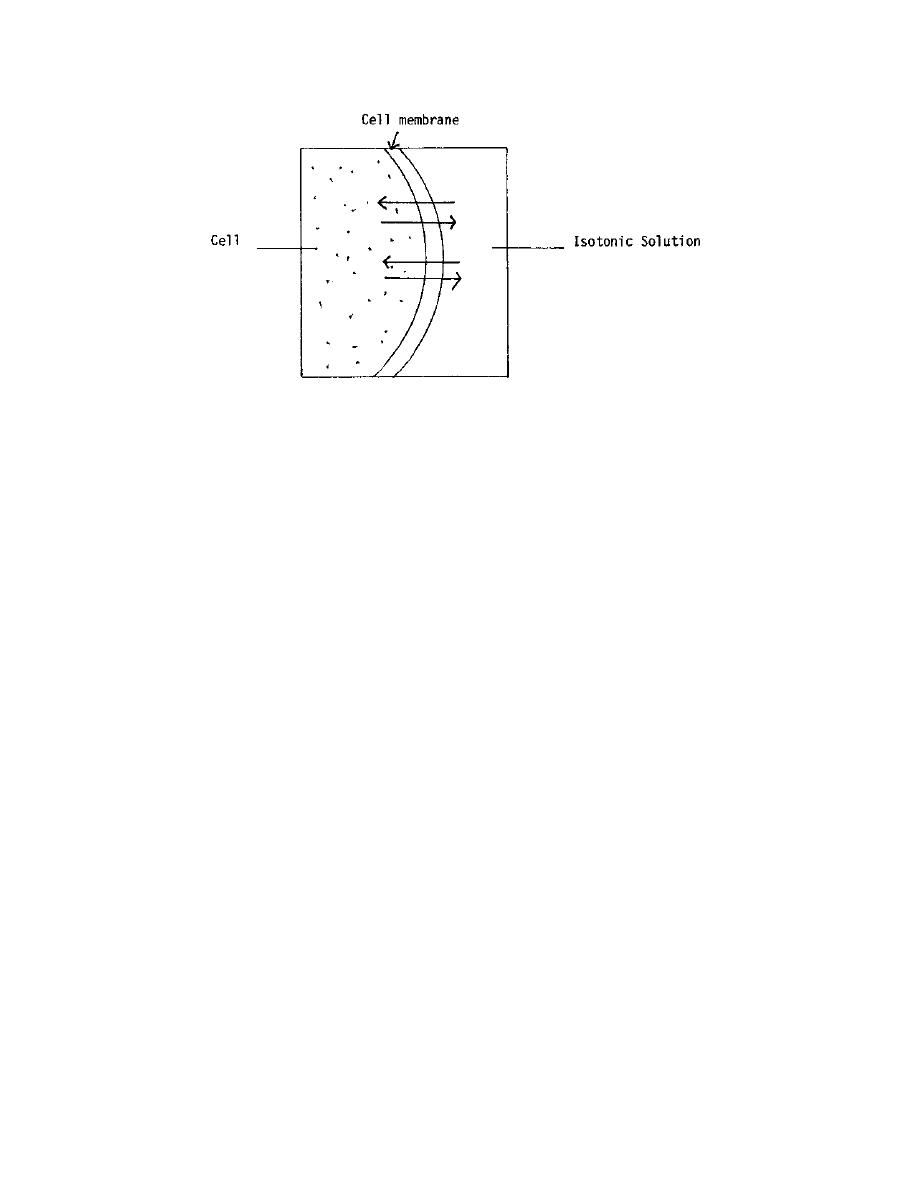
Figure 2-6. Isotonic solution.
2-8.
DIFFUSION AND OSMOSIS
Even though the predominate movement of hypotonic solutions and hypertonic
solutions is in one direction, there is always a weak movement in the opposite direction.
This weak movement is caused by diffusion. This process is in contrast to osmosis,
which is a directional movement.
a. Diffusion. When a solute is added to a fluid, the molecules will begin
immediately to spread throughout the fluid. This process is diffusion. The movement is
random. It is the nature of molecules to move constantly in a fluid. Even though no
special effort (like stirring) is made to mix the solution, the solute will be evenly
distributed within a period. An illustration of diffusion is to add ice cubes to a warm
liquid. Without stirring, the cold molecules will distribute themselves among the warmer
ones. This will create a liquid of uniform temperature. The diffusion process is not
limited by the presence of a semi-permeable membrane as long as the molecules are
small enough to travel through the membrane. Cell walls are semi-permeable
membranes.
b. Osmosis. The process of osmosis is unidirectional across a semi-permeable
membrane. When the molecules of a solute are too large to travel through the
membrane wall, they will remain on one side. When a solvent is added to the other side
of the membrane, the molecules of the solvent will travel through the membrane to the
side with the greater concentration of the solute. This is the principle by which the
hypotonic and hypertonic solutions move fluid across the cell membranes.
2-9.
CRYSTALLOID AND COLLOID SOLUTIONS
The nature of injectable crystalloid and colloid solutions determines their ability to
be absorbed by the cells or to remain in the circulatory system.
MD0564
2-9



 Previous Page
Previous Page
