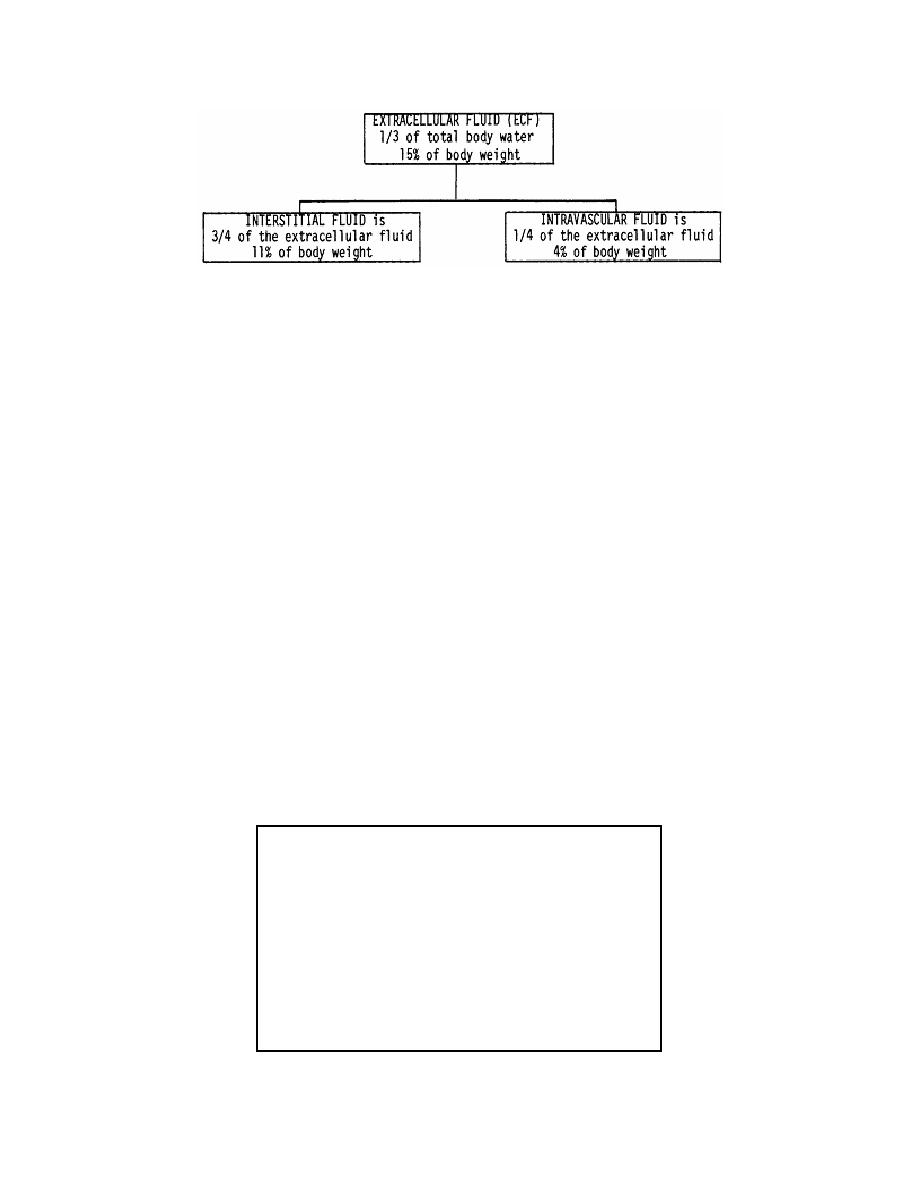
Figure 2-2. Extracellular fluid distribution.
Section II. ELECTROLYTES IN BODY FLUIDS
2-5.
ELECTROLYTES
The amounts of intracellular and extracellular fluids contained in a person's body
are extremely important to his healthy physiology. Losses of body fluids by vomiting,
diarrhea, or excessive perspiration can produce illness or even death. Whenever body
fluids are lost, the substances dissolved in the fluids are also lost. Certain inorganic
substances are found in the body's fluids. These are called "electrolytes." Examples of
electrolytes are potassium and chloride. These electrolytes exist in their "ion" state in
the body fluids. This means that each ion can combine with one or more ions to form
needed body compounds or can produce electro-chemical equilibrium (or balance).
One example of this is the osmotic pressure that causes water to flow across a cell
membrane. The relationship between the concentrations of sodium and potassium
electrolytes in the cells and the extracellular fluid causes the water to flow into and out
from the cells. There is usually a low level of sodium in the cells and a high
concentration of potassium. The milliequivalent (mEq) is a unit of measure for the
electrolyte.
a. The movement of electrolytes is governed by their electrical charge. Some
are positively charged and are called "cations." Others are negatively charged and
called "anions." Below are the major electrolytes, their chemical abbreviations, and the
amount of each contained in a liter of extracellular fluid.
+
Sodium (Na )
140 mEq
Chloride (Cl )
100 mEq
Bicarbonate (HCO3 )
27 mEq
+
Potassium (K )
4 mEq
+
(Mg 2)
Magnesium
3 mEq
MD0564
2-4



 Previous Page
Previous Page
