(a)
Liquid petrolatum (mineral oil). This liquid is used as a solvent and
as a laxative.
(b)
Petrolatum (petroleum jelly). This semisolid is used as an ointment
base.
(c)
Paraffin (wax). This solid is used in pharmacy as a stiffening agent.
(2) Unsaturated aliphatic hydrocarbons. The second type of aliphatic
hydrocarbon is unsaturated hydrocarbons. These are hydrocarbons, which contain at
least one double or triple bond (that is, they are alkenes or alkynes). An example of an
alkene is ethene, the simplest alkene, which consists of two double-bonded carbon
atoms and four hydrogen atoms.
CH2 = CH2
Note that the name is similar to the saturated compound ethane. The -ene ending
comes from the word alkene and denotes that it contains a double bond. Similarly, if
there were a triple bond between the two carbon atoms, the name would be ethyne with
the -yne ending denoting the triple bond (from alkyne). The physical properties of
alkenes and alkynes are similar to the properties of alkanes of similar molecular
weights. Chemically, the word unsaturated implies that these compounds can form
additional bonds. This is the case, for alkenes and alkynes are much more reactive and
undergo many reactions not possible with alkanes.
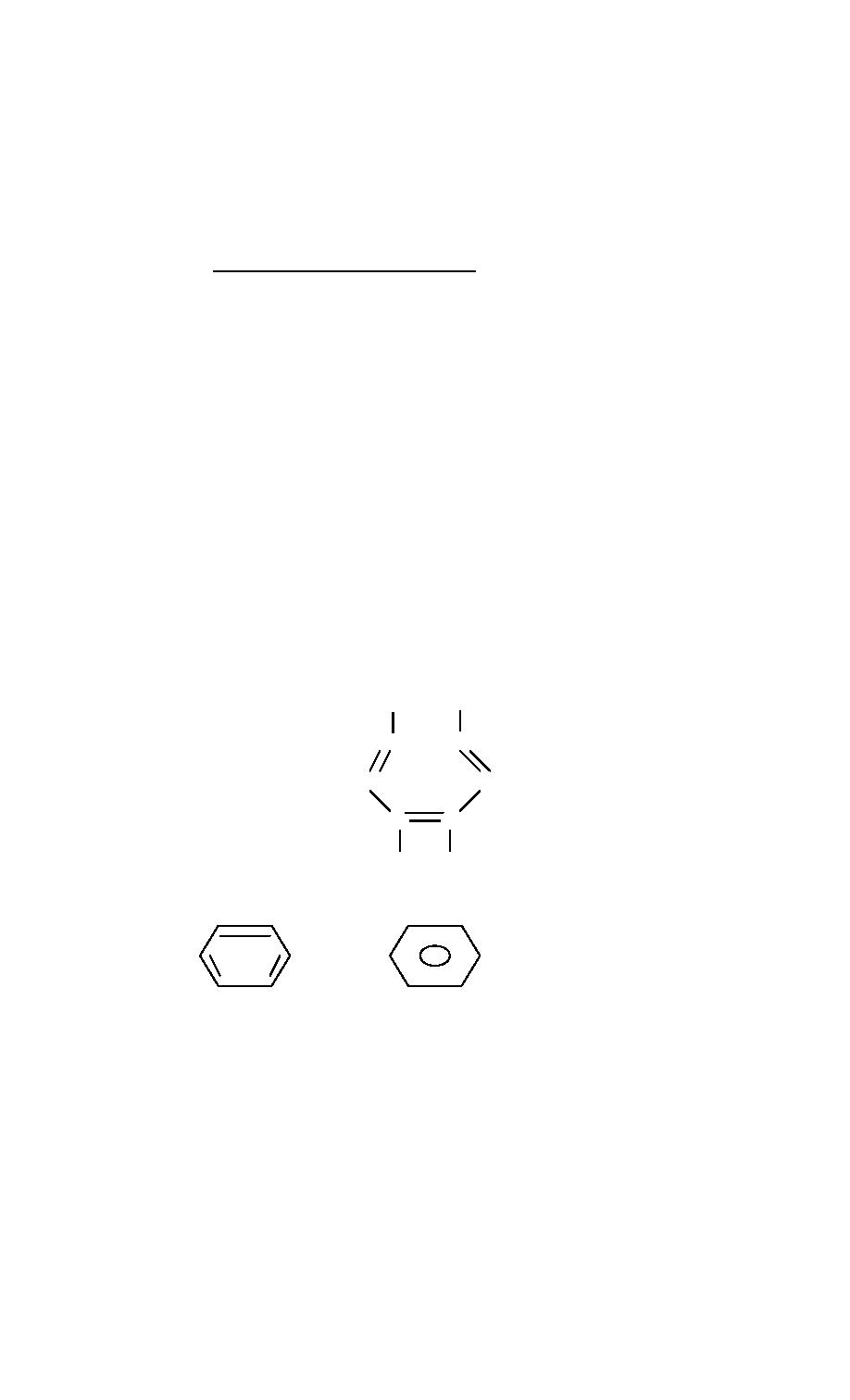
b. Aromatic Hydrocarbons. The second major group of the hydrocarbons is
the aromatic hydrocarbons, which are hydrocarbons that contain a benzene ring as part
of their structure. Benzene has the formula C6H6 and consists of six carbon atoms in a
ring with three alternating double bonds.
H
H
C
C
H-C
C-H
C
C
H
H
The benzene ring is also represented with the following symbols:
or
or
3-6
MD0803




 Previous Page
Previous Page
