(4) One reason the oxidation reaction is important is that it is the means the
body uses to eliminate the popular liquid, ethyl alcohol, or ethanol (CH3-CH2-OH).
c. Uses of Alcohols. Alcohols are most commonly used as solvents in the
pharmacy. They are also used as disinfectants and antiseptics.
3-9.
PHENOLS
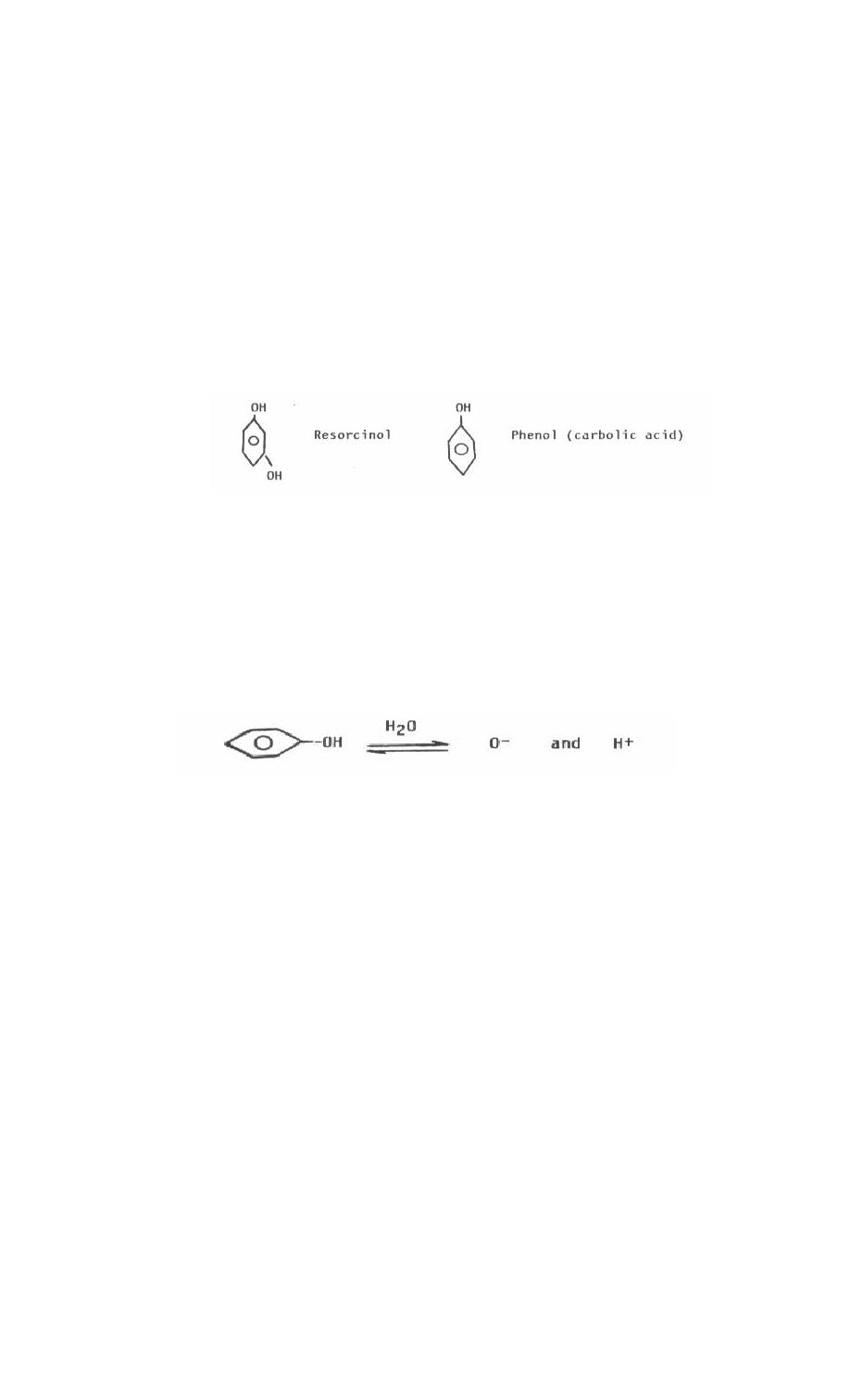
Phenols are hydroxyl derivatives of hydrocarbons formed by replacing a
hydrogen on the benzene ring of an aromatic hydrocarbon with the hydroxyl radical.
Phenols have the general formula Ar-OH, where Ar represents a substituted or
nonsubstituted aromatic hydrocarbon. Thus, phenols are really just a special class of
alcohols. However, they have enough unique properties that they deserve to be
considered as a separate class of compounds. Below are some examples of typical
phenols.
a. Properties of Phenols. All phenols are white solids with moderately high
melting points and are soluble in water. They also have the property of being able to
form eutectics with camphor, menthol, or thymol, which are solid alcohols. (A eutectic is
a uniform mixture formed from two compounds that melt at a temperature lower than the
melting point of either of the two compounds.) Thus, phenol (a solid) and camphor (a
solid) form a liquid mixture at room temperature which is called a eutectic.
b. Reactions of Phenols. Chemically, phenols are weakly acidic compounds.
The hydrogen dissociates to a small degree from the hydroxyl radical to act as an acid
as shown below.
Since phenols are weak acids, they will form salts with inorganic bases. Phenols with
two hydroxyl groups also undergo oxidation reactions.
c. Uses of Phenols. Medicinally, phenolic compounds have three uses: as
keratolytics (compounds that remove hornified or scaling outer layers of skin),
antipruritics (relieve itching), and disinfectants. These uses arise from the fact that
phenols are very caustic to animal tissues. Precautions must therefore be taken when
you are using phenols in preparations. These properties, possessed to different
degrees by various phenols, depend on which other functional groups are present and
the number of hydroxyl groups.
3-10
MD0803



 Previous Page
Previous Page
