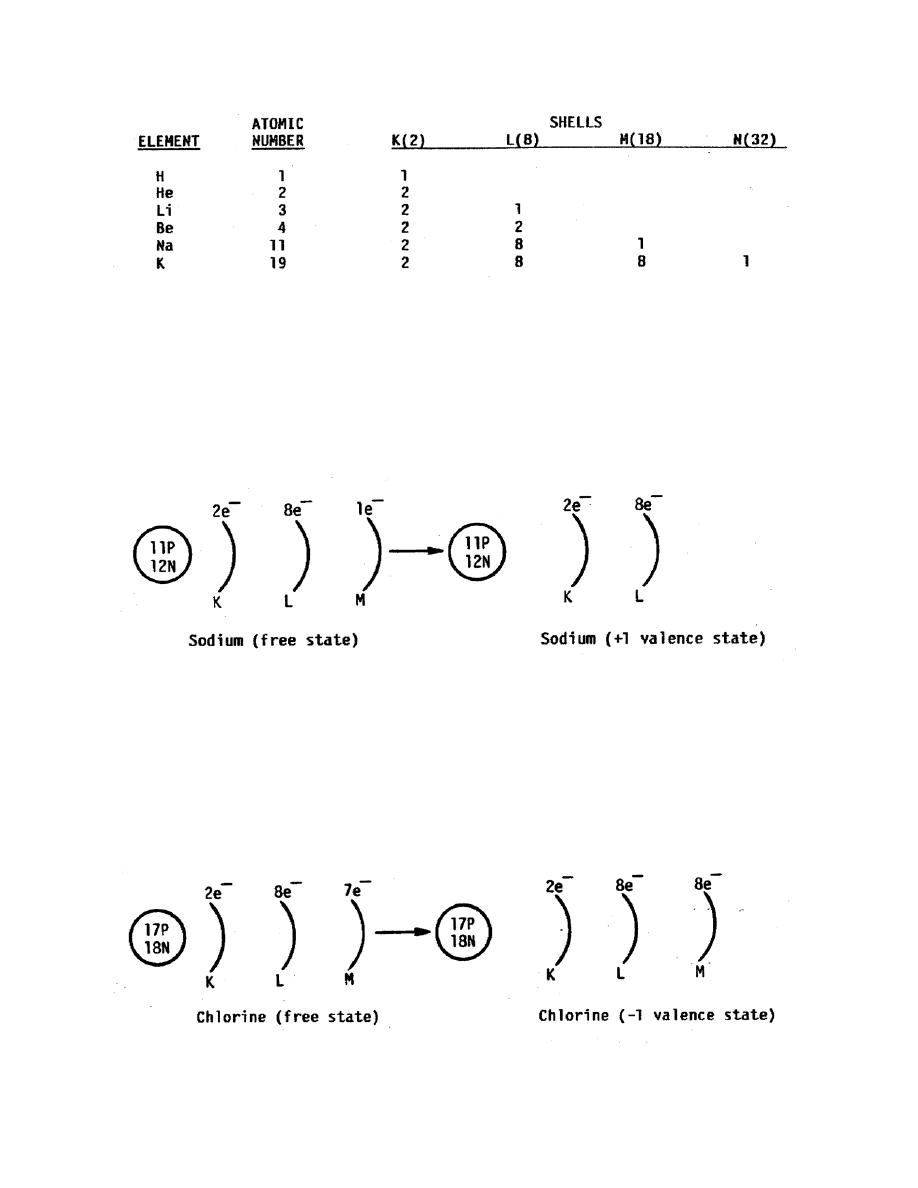
c. Positive Valence. An atom that must give up electrons to become stable will
have more protons than electrons in its stable configuration and will not be electrically
neutral. It will be positively charged since there are more positive charges than
negative charges. This is indicated by a + sign. The number of electrons it gives up is
the numerical value of its valence. Consider, for example, the element sodium, which
has 11 protons and 11 electrons in its free state. It has one electron in the M shell,
which it loses easily to become stable. After it loses the electron (that is, gives up a
negative charge), it will have a positive one charge and its valence will be +1.
d. Negative Valence. An atom that must gain electrons to become stable will
have more electrons than protons in its stable configuration and will not be electrically
neutral. It will be negatively charged since there are more negative than positive
charges. This is indicated by a "-" sign. The number of electrons it gains is the
numerical value of its valence. Consider, for example, the element chlorine, which has
17 protons and 17 electrons in its free state. It is one electron short of fitting the octet
rule in the M shell as that shell contains 7 electrons. After it gains the electron, it will
have a negative one charge and its valence will be -1.
MD0803
1-14



 Previous Page
Previous Page
