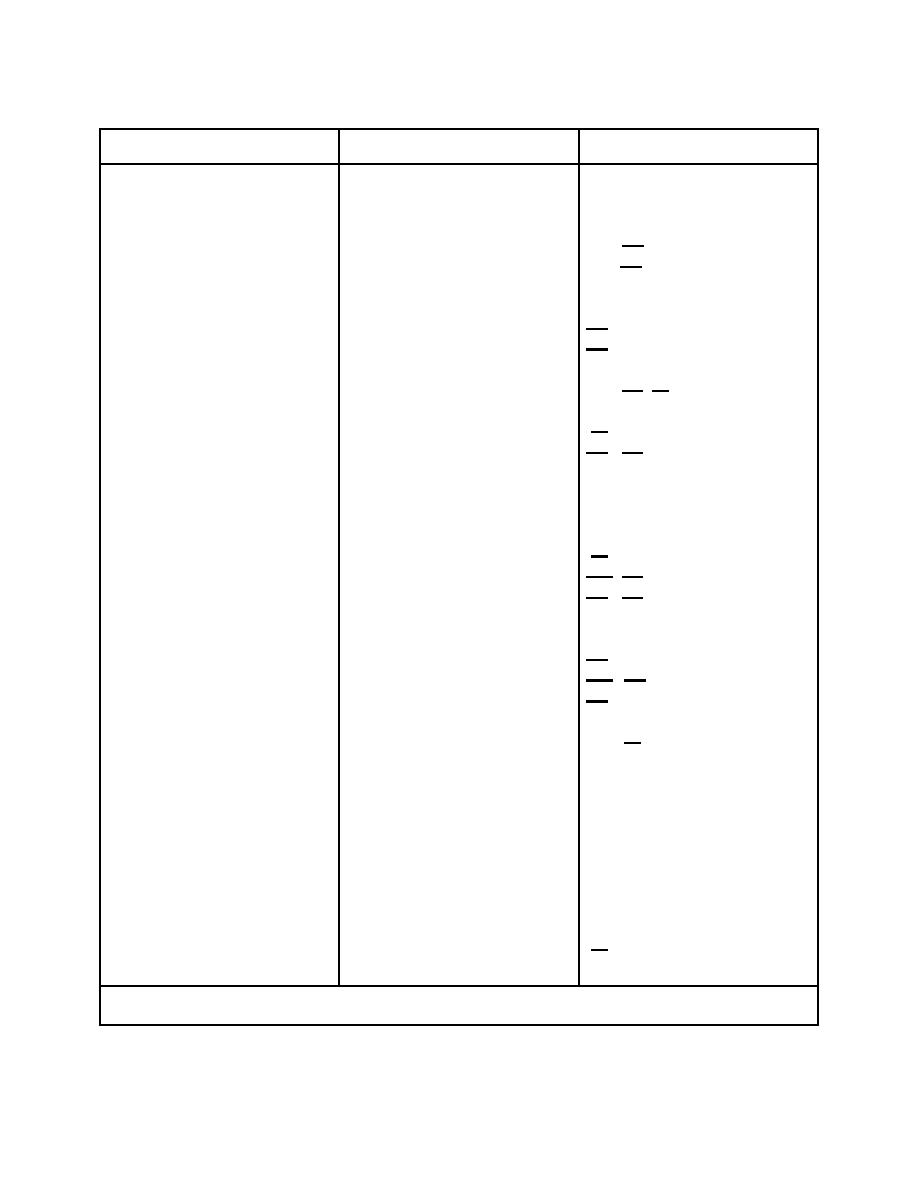
NAME
SYMBOL
VALENCE
Acetate
C2H3O2
-1
Aluminum
Al
+3
Ammonium
NH4
+1
Antimony
Sb
-3,
+3, +5
Arsenic
As
-3,
+3, +5
Barium
Ba
+2
Bicarbonate
HCO3
-1
Bismuth
Bi
+3,
+5
Bromine
-1,
Br
+1, +3, +5, +7
Calcium
Ca
+2
Carbon
C
+2,
+4, -4
Carbonate
CO3
-2
Chlorine
-1,
Cl
+1, +3, +5, +7
Copper
Cu
+1,
+2
F
-1
Gold
Au
+1,
+3
H
+1
Hydroxide (Hydroxyl)
OH
-1
Iodine
I
-1,
+1, +3, +5, +7
Iron
Fe
+2,
+3
Lead
Pb
+2,
+4
Lithium
Li
+1
Magnesium
Mg
+2
Mn
+2,
+3, +4, +6, +7
Mercury
Hg
+1,
+2
Nickel
Ni
+2,
+3
Nitrate
NO3
-1
Nitrogen
+1,
-3, +3, +5
N
Oxygen
-2
0
Permanganate
-1
MnO4
Phosphate
-3
PO4
Phosphorus
-3,
+3, +5
P
Potassium
+1
K
Silver
+1
Ag
Sodium
+1
Na
Strontium
+2
Sr
Sulfate
-2
SO4
Sulfur
-2,
S
+2, +4, +6
Zinc
Zn
+2
NOTE: The most common valences are underlined where there may be more than one
valence.
Table 1-3. Valences.
MD0803
1-17



 Previous Page
Previous Page
