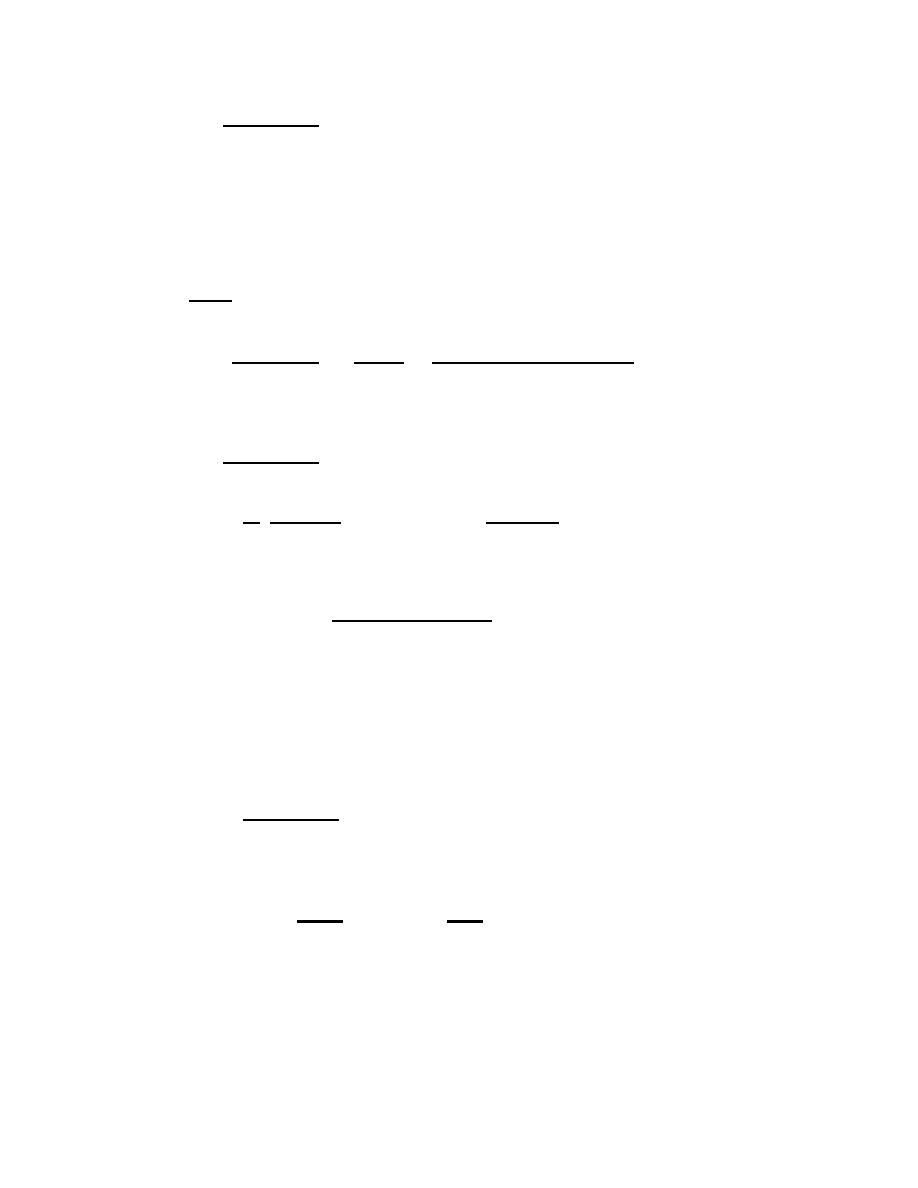
(2) Example #2:A 0.5% (v/v) glacial acetic acid solution would contain 0.5
milliliters of glacial acetic acid in each 100 milliliters of solution.
d. When the type of percent is not stated, it is understood that dilutions of (1)
dry ingredient in a dry preparation are percent W/W, (2) dry ingredients in a liquid are
percent W/V, and (3) a liquid in a liquid is percent V/V.
2-6. METHODS FOR SOLVING PERCENTAGE PROBLEMS
a.
Ratio and proportion:
Formula:
IF # of parts = THEN
Amount of solute needed
100
Total volume or weight of product
b.
Sample problems:
(1) Example #1:How many grams of zinc oxide are needed to make 240
grams of a 4% (w/w) zinc oxide ointment?
IF 4 g ZnO =
THEN
X g ZnO
100 g Oint
240 g Oint
NOTE: Because all the units involved in this problem are the same (grams), each unit
is labeled as to what it represents. In the first ratio, the desired strength of the ointment
is indicated. A four percent zinc oxide ointment contains 4 grams of zinc oxide in each
100 grams of ointment and is labeled to indicate this. Because the question asks, "how
many grams of zinc oxide?," the X value must be placed opposite the grams of zinc
oxide in the first ratio (see above). The 240 grams of ointment is placed opposite the
grams of ointment in the first ratio.
Then: 100 X = 960
X = 9.6 g of zinc oxide
(2)
Example #2: How many milliliters of a 5% (w/v) boric acid solution can
be made from 20 grams of boric acid?
IF
THEN
5g
=
20 g
100 ml
X ml
5X
=
2000
X
=
400
MD0802
2-13



 Previous Page
Previous Page
