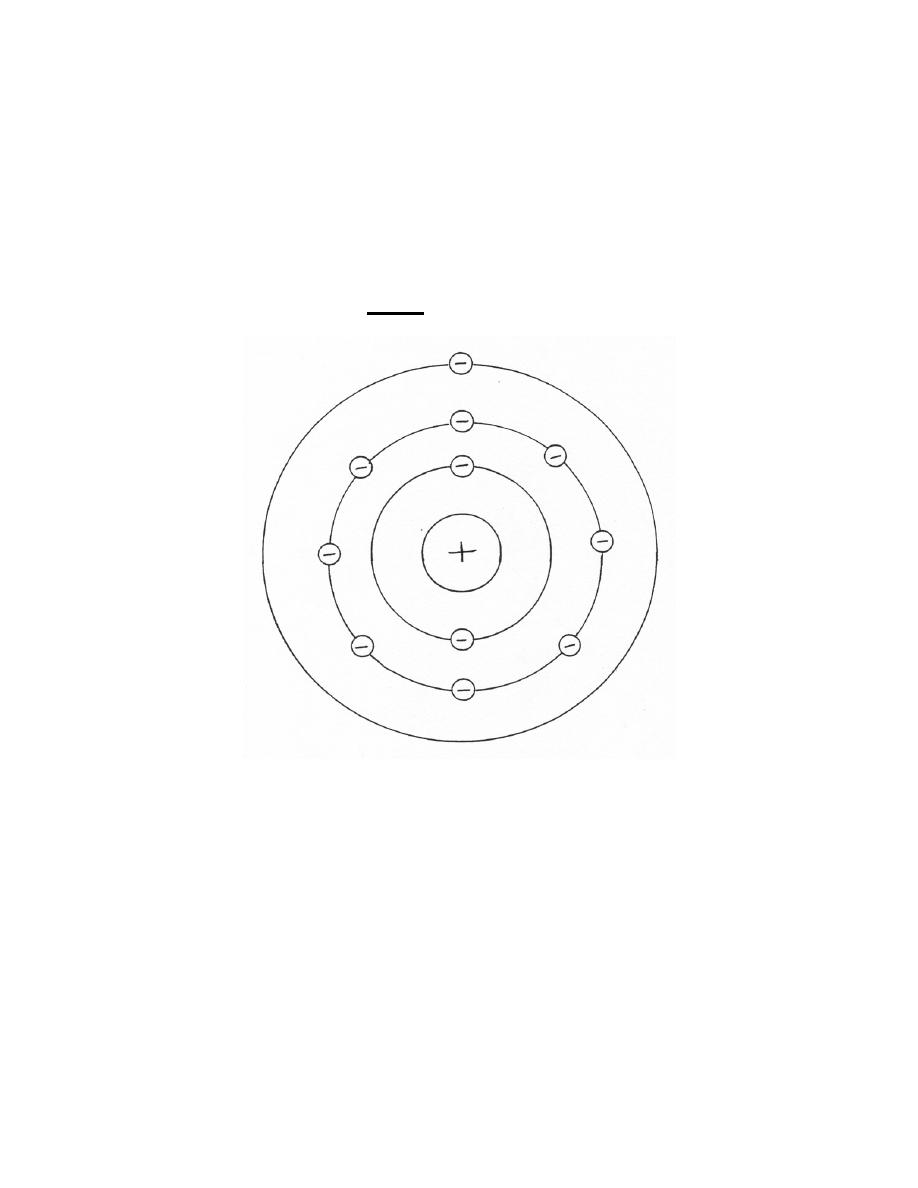
b. Bohr's Theory. Different theories have been advanced and improved upon
over the years to explain the structure of the atom. Bohr's theory, proposed in 1913,
likens the atom to a miniature solar system (figure 1-2). According to this concept, a
positively charged nucleus containing protons (positive particles) and neutrons (neutral
particles) is located in the central core. It contains nearly all the mass (weight) of the
atom. Revolving around the nucleus are almost weightless orbital electrons--planetary
particles with negative charges. In the stable state (called the neutral state), these
negative charges are balanced against the positive charges in the nucleus. Once this
balance in atomic charge is disrupted (by the loss or gain of electrical charge, for
instance), the atom is said to be ionized.
Figure 1-2. Atomic model as envisioned by the Bohr theory.
c. Wave Theory. The newer and more accepted theory of atomic structure
based upon wave mechanics treats electrons as a kind of three-dimensional cloud
spread around the nucleus as electron waves. Even though Bohr's concept of the atom
with sharply defined electron orbits is now replaced by the electron cloud theory, the
equation developed by Bohr on electron activity generally agrees with experimental
observations. Therefore, for our purposes, the Bohr orbit concept will be used as it
lends itself more effectively to visualization. Though the new electron wave theory may
explain all known phenomena more satisfactorily, it is difficult to form a mental picture of
what an atom might look like.
MD0950
1-8



 Previous Page
Previous Page
