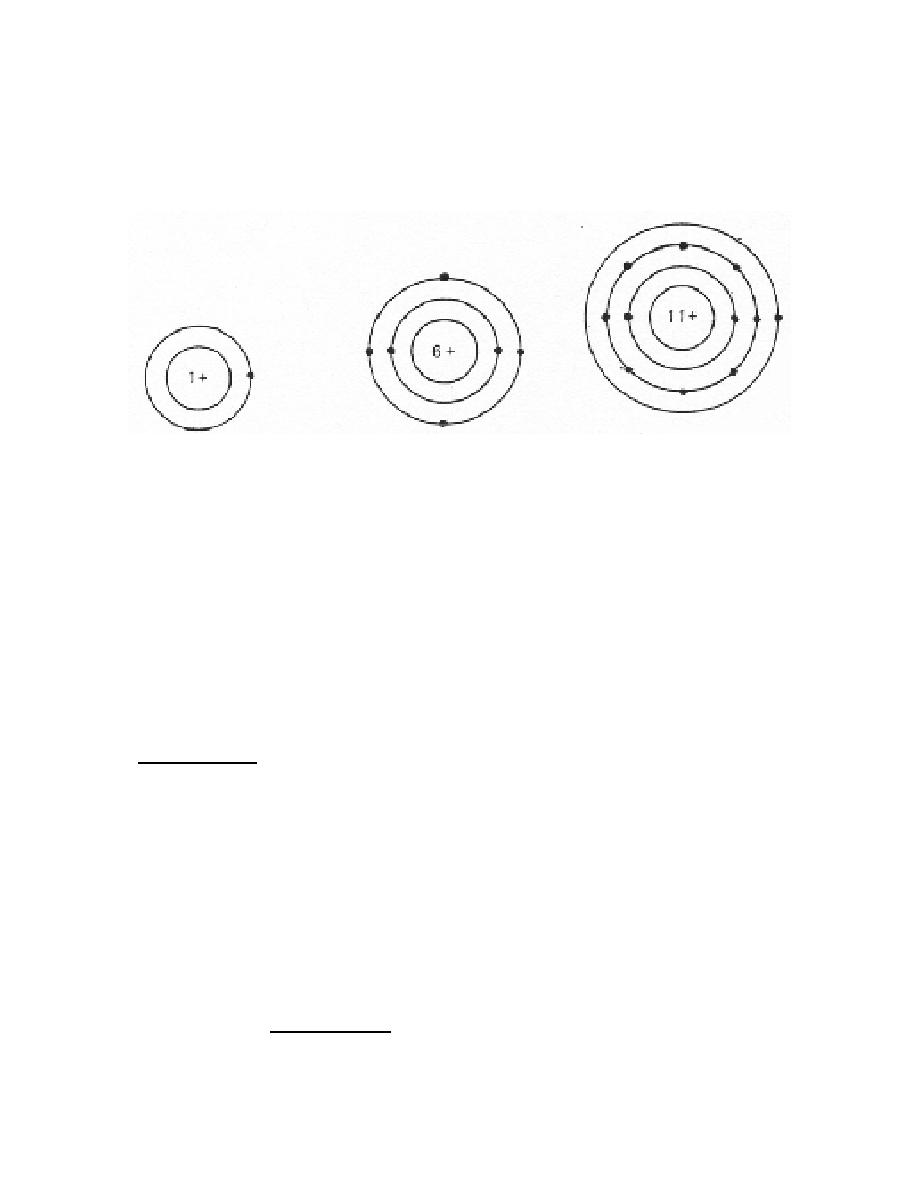
proton) has a single orbiting electron. The carbon atom with a nucleus containing six
protons has two "shells" of orbiting electrons, with two electrons in the inner and four in
the outer energy level. Also, notice that the sodium atom has 11 protons in its nucleus
and three shells of orbiting electrons with a single electron in its outermost shell.
Hydrogen atom
Carbon atom
Sodium atom
1
2, 4
2, 8, 1
(5) The use of orbits and shells to describe electron position indicates the
relationship of different electron-energy levels. Generally speaking, an atom will tend to
react with other elements more readily when its outer shell is not completely filled. This
concept of interaction of elements will be further discussed later in this lesson under
valence and molecule formation.
(6) Electrons are maintained in their shells by a combination of centrifugal
force (which tends to propel them away from the nucleus) and electrostatic force, and
the attraction between the positive nucleus and negative electrons. Since the electrons
are bound to the nucleus by electrostatic force, a certain amount of energy is required to
remove an electron from its shell to a point completely outside the atom. This is called
the binding energy of that shell. The binding energy of the shell of an atom is greatest
in the K shell and decreases as the distance of the shell from the nucleus increases.
For example, the binding energies for tungsten shells are: K--69.5 keV, L--12 keV,
M--2 keV, and N--0.8 keV. Consequently, it would require at least 69.5 keV of energy in
an atom of tungsten to remove a K electron from its shell, at least 12 keV to remove an
L electron from its shell, and so on. The binding energy is important in the production of
characteristic radiation that is explained later in this lesson.
(7) The binding energy of a shell, and consequently of an electron in that
shell, must not be confused with the potential energy possessed by that electron. The
potential energy possessed by the electrons is least in the K shell and increases with
distance from the nucleus.
(a) Binding energy. To illustrate the difference between binding energy
and potential energy of the electrons, we will compare an atom to a metal ball and
magnet. Suppose the ball was permitted to occupy three steps at different distances
MD0950
1-10



 Previous Page
Previous Page
