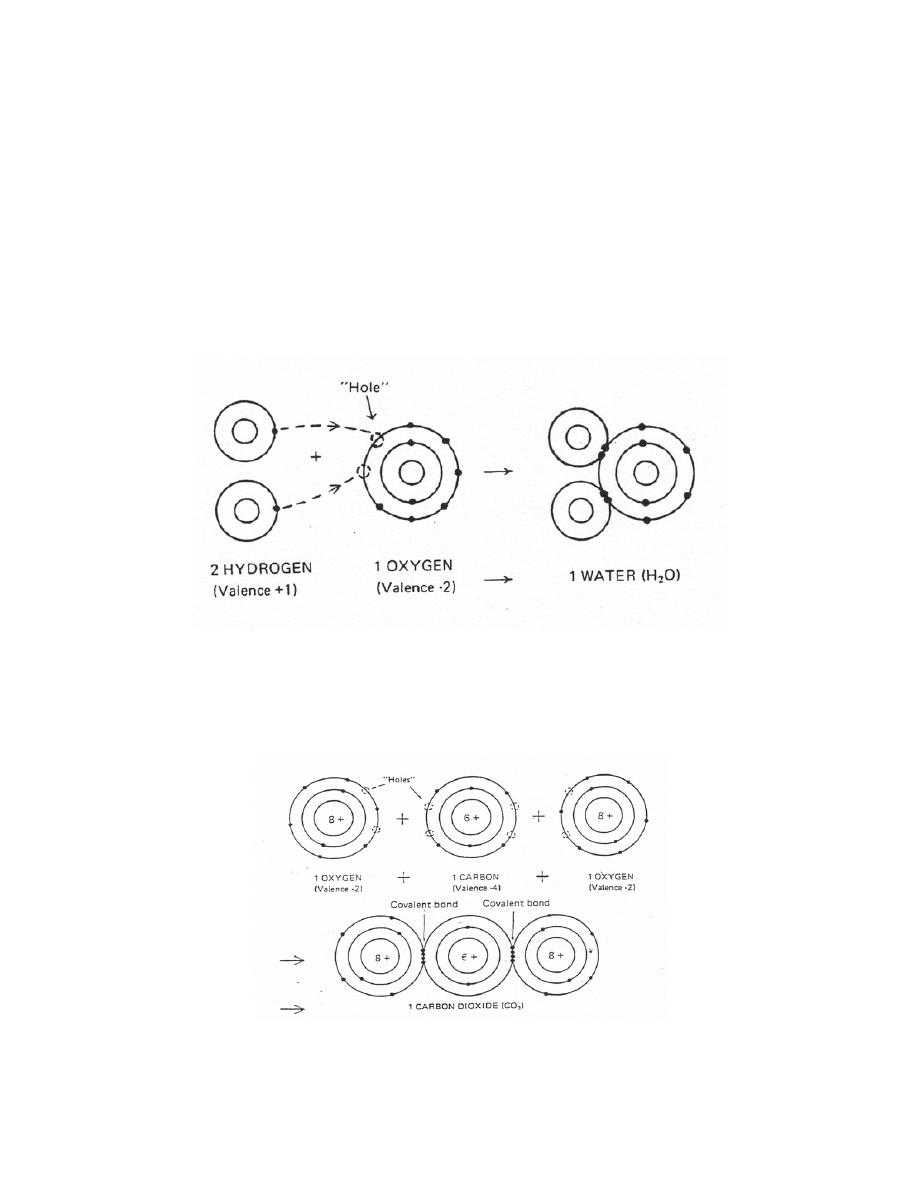
b. Convalent Bond. In contrast, a convalent bond is formed by the sharing of
valence electrons between atoms.
(1) A water molecule is an example (figure 1-8). Here, one oxygen atom is
linked to two hydrogen atoms by the single electrons of the two hydrogen atoms to form
a water molecule. Actually, the single electron from each hydrogen atom is paired with
a single electron from the oxygen atom in this covalent relationship. For part of the
time, each paired electron can be considered as circulating around the hydrogen
nucleus and, for the rest of the time, around the oxygen nucleus. (Note: Slightly more
time is spent around the electronegative oxygen atom.) By doing so, both the oxygen
and hydrogen atoms have completed shells and established a more stable
configuration.
Figure 1-8. Covalent bond - water molecule.
(2) Another example is a molecule of carbon dioxide, which illustrates the
double covalent bond (figure 1-9). Here two oxygen atoms are linked by the four
electrons they share at each juncture with the carbon atom.
Figure 1-9. Double convalent bond carbon dioxide molecule.
MD0950
1-14



 Previous Page
Previous Page
