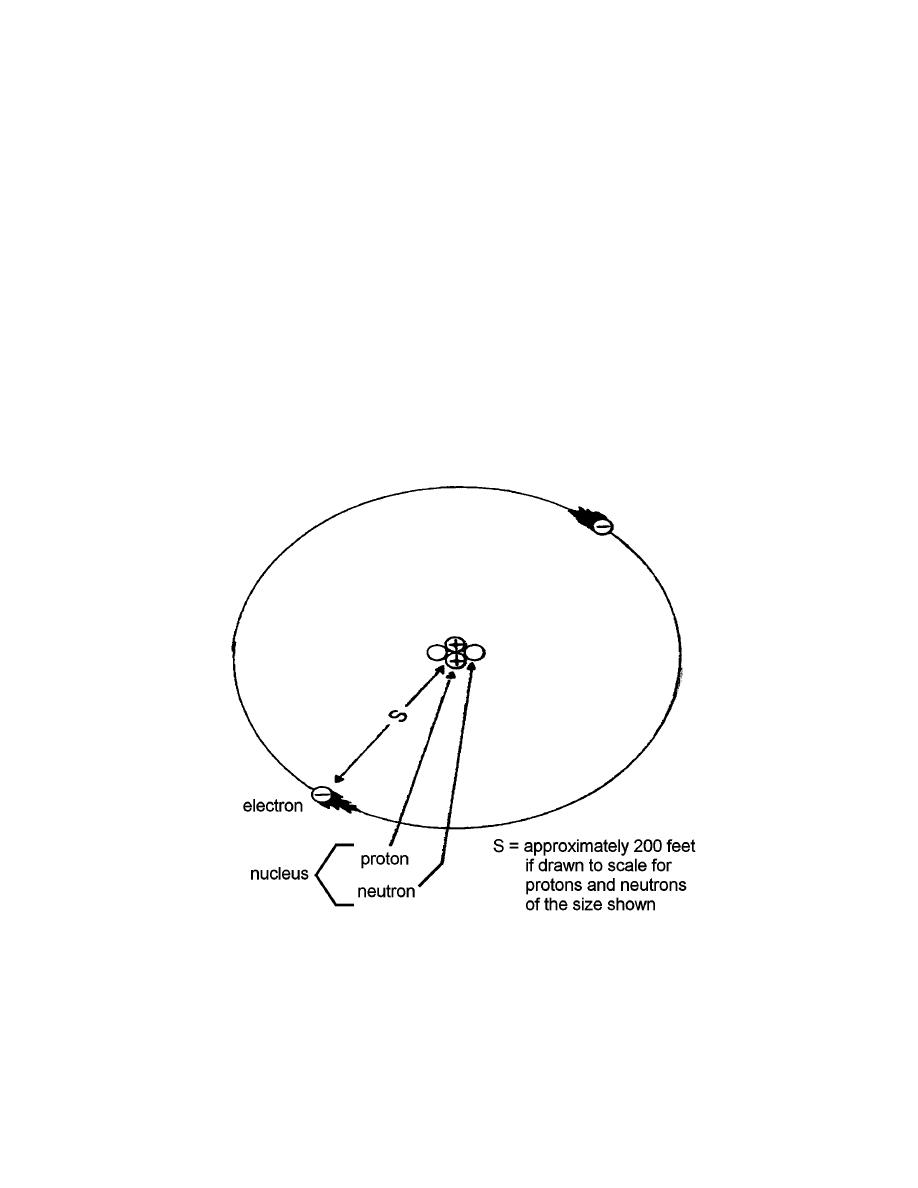
e. There was still the problem of determining how these basic particles are
arranged to make up an atom. It has been learned that atoms are composed of a
positively charged central mass called the nucleus (which contains protons and
neutrons) and electrons which move in orbits or shells around, but very far from, this
nucleus. It is reasonable to assume the electrons experience an attractive force due to
the positive nucleus. However, they move rapidly enough so that the centrifugal force
(tending to throw the electrons out of orbit) balances the attractive force. Most objects
in the world have no charge on them; therefore, we can reasonably suppose that the
atoms of which matter is composed are electrically neutral, that is, they have no net
charge. Thus, atoms normally contain exactly as many electrons moving in shells
around the nucleus, as there are protons in the nucleus. There are no electrons as
electrons. Thus, because the electrons are very small in mass and since they move
around the nucleus at distances relatively far from the nucleus, the atom is primarily
empty space with the major portion of its mass concentrated in the nucleus. An atom is
about 10-8 centimeters in diameter; this essentially refers to the diameter of the electron
orbits, the nucleus being about 10-12 centimeters in diameter (Figure 1-1).
Figure 1-1. Typical simple atom.
f. Neutrons, being neutral in charge, do not affect the chemical nature of the
atom and it is the number of protons in the nucleus that determines the element to
which the atom belongs. For example, an atom of the lightest element, hydrogen, has
one proton in the nucleus and one electron traveling around the nucleus. An atom
MD0180
1-5



 Previous Page
Previous Page
