containing two protons in the nucleus and two electrons traveling around the nucleus
belongs to the element helium. Similarly, the 92d natural element, uranium, is
composed of atoms containing 92 protons in the nucleus and 92 electrons traveling in
shells around the nucleus. The number of neutrons in the nucleus of atoms of any
particular element varies, but there are usually more neutrons than protons in the
nucleus of an atom. Two atoms of the same element with different numbers of neutrons
are called isotopes.
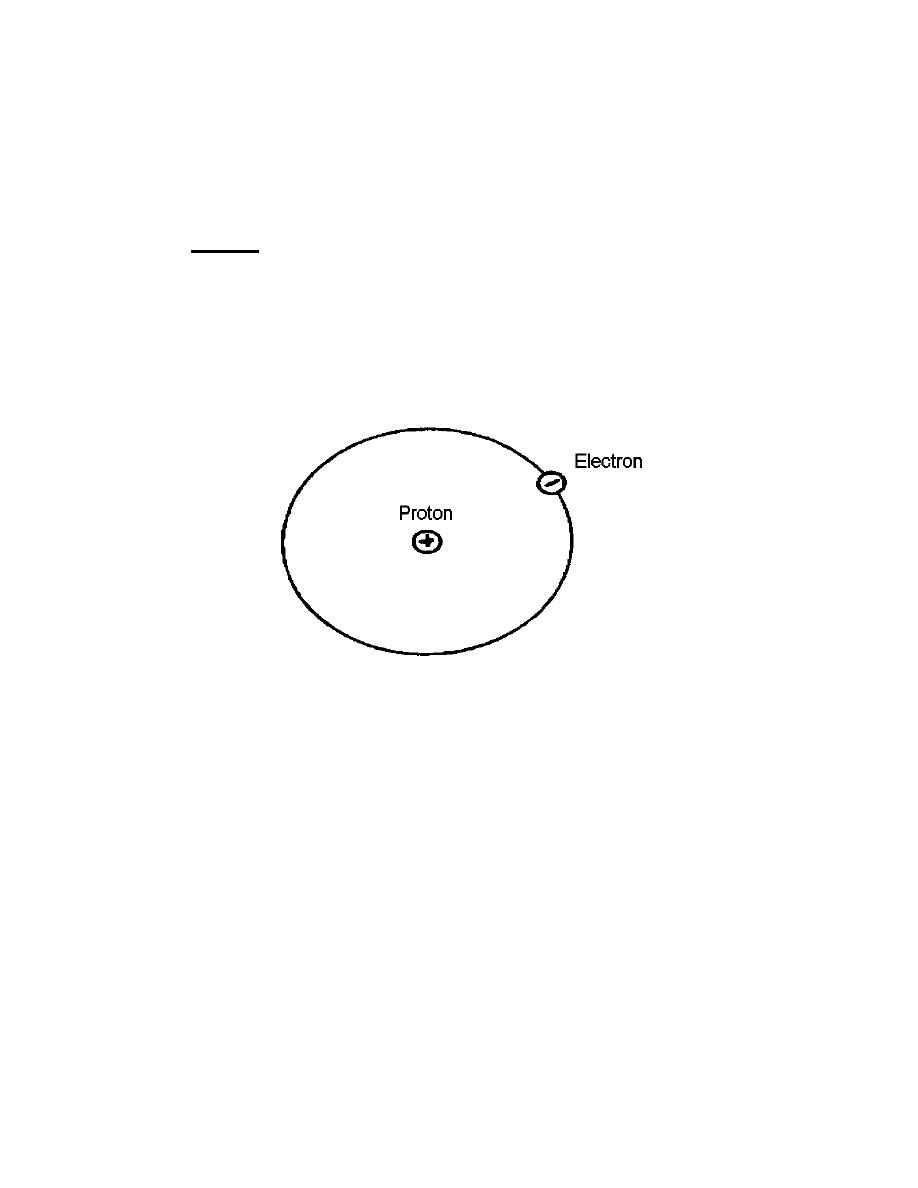
g. Electrons are not distributed at random about the nucleus, but they exist in
arrangements that follow definite laws. The model of the atom proposed by Niels Bohr
in 1913 pictures the electrons as moving in circular orbits about the nucleus. Although
we now know this model is not strictly correct, its features give a good explanation of
the simple phenomena, which we wish to consider. Figure 1-2 shows a picture of the
hydrogen atom.
Figure 1-2. Hydrogen atom.
h. The next heaviest element after hydrogen is helium. This contains two
protons in the nucleus and, consequently, two electrons revolving about the nucleus
(Figure 1-3). Both these electrons may be thought of as roughly the same distance
from the nucleus. It turns out that no more than two electrons will ever be found at this
distance from the nucleus in helium. Of course, this is not unique with helium, but is
also true of elements with more than two protons in the nucleus. Both these electrons
are said to lie in a shell, which is actually an orbit around the nucleus. No more than
two electrons may be present in the shell, no matter what atom is under consideration.
Normal hydrogen has only one electron in the shell, so we call the shell incomplete.
i. Atoms which have more than two protons in the nucleus have an equal
number of electrons; in this case, the electrons in excess of two will lie in shells past the
first one. The shells are normally designated by capital letters, the first shell being
denoted by "K," the second by "L," the third by "M," etc. It is possible that one of the
electrons revolving about a helium nucleus might be in the K-shell and the other in the
L-shell. This puts only one electron in the K-shell and the K-shell can hold two. Such a
MD0180
1-6



 Previous Page
Previous Page
