e. Periodic Law. While investigating the properties of the elements, scientists
discovered an interesting fact that is now called the periodic law. This law states that
the properties of the elements are periodic functions of the atomic number. As the
atomic number increases, the properties of the elements repeat themselves at regular
Intervals.
f. Periodic Table. The periodic law allowed the scientists to group together the
elements that had similar properties and form a systematic table of the elements. This
table is the periodic table (Table 1-2). The vertical columns are called groups, and the
horizontal rows are called periods. This table contains a lot of information that we will
not generally use; however, we are concerned with the basic information we can obtain
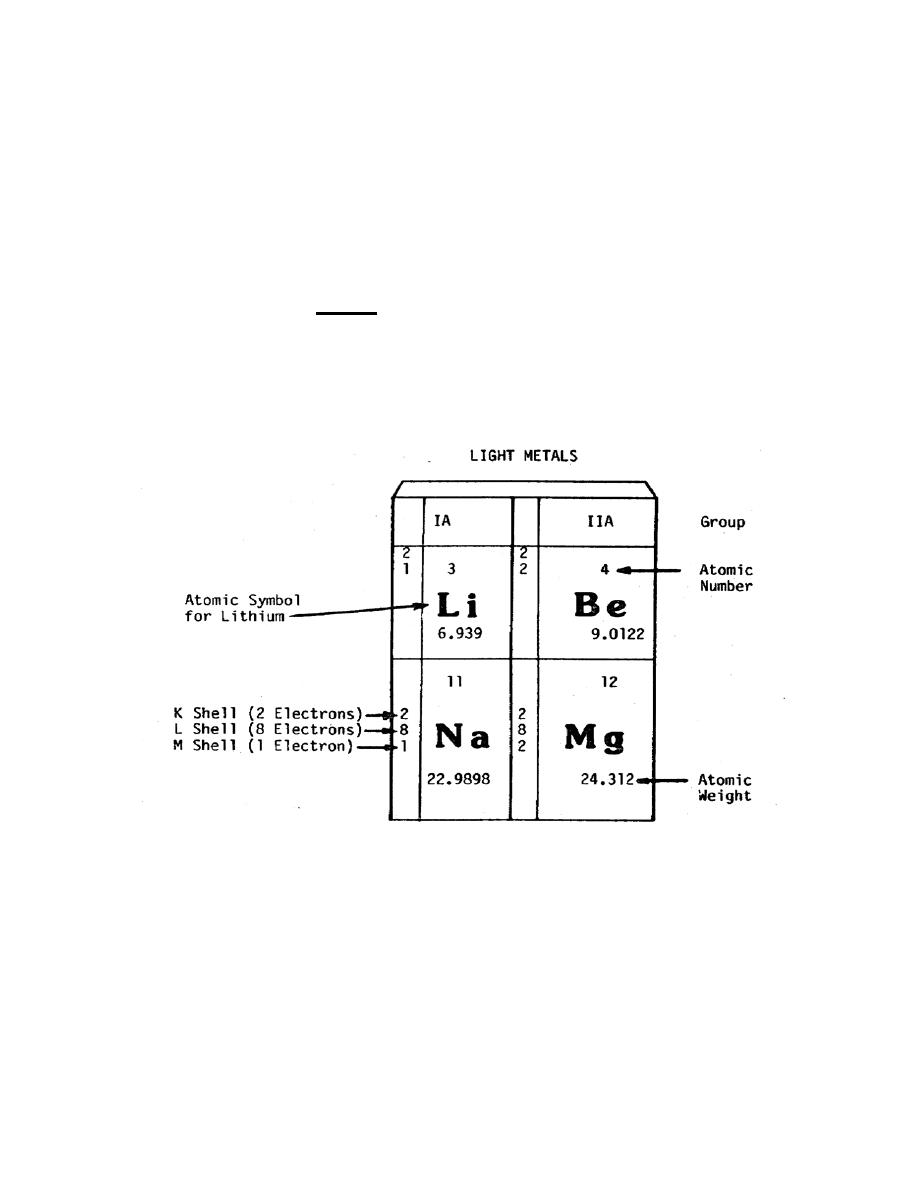
about the elements. Figure 1-2 includes four blocks for elements from the periodic table
showing the information, which can be obtained from it. You should note that the
number of neutrons is not given in the periodic table. This can be determined by
subtracting the atomic number from the atomic weight.
Figure 1-2. Identifying the components of the periodic table.
g. Isotopes. All the atoms of a particular element are not identical. Slight
variations in the number of neutrons are found to occur naturally. Variations can also
be produced in reactors. Atoms that have the same number of protons but a different
number of neutrons (same atomic number, but different atomic weights) are called
2
3
239
isotopes. Sometimes isotopes are referred to by their mass numbers, H , H , U , and
so forth. All of the isotopes of a particular element have identical electronic
configurations; and since electronic configurations determine chemical properties,
isotopes of an element
MD0803
1-11



 Previous Page
Previous Page
