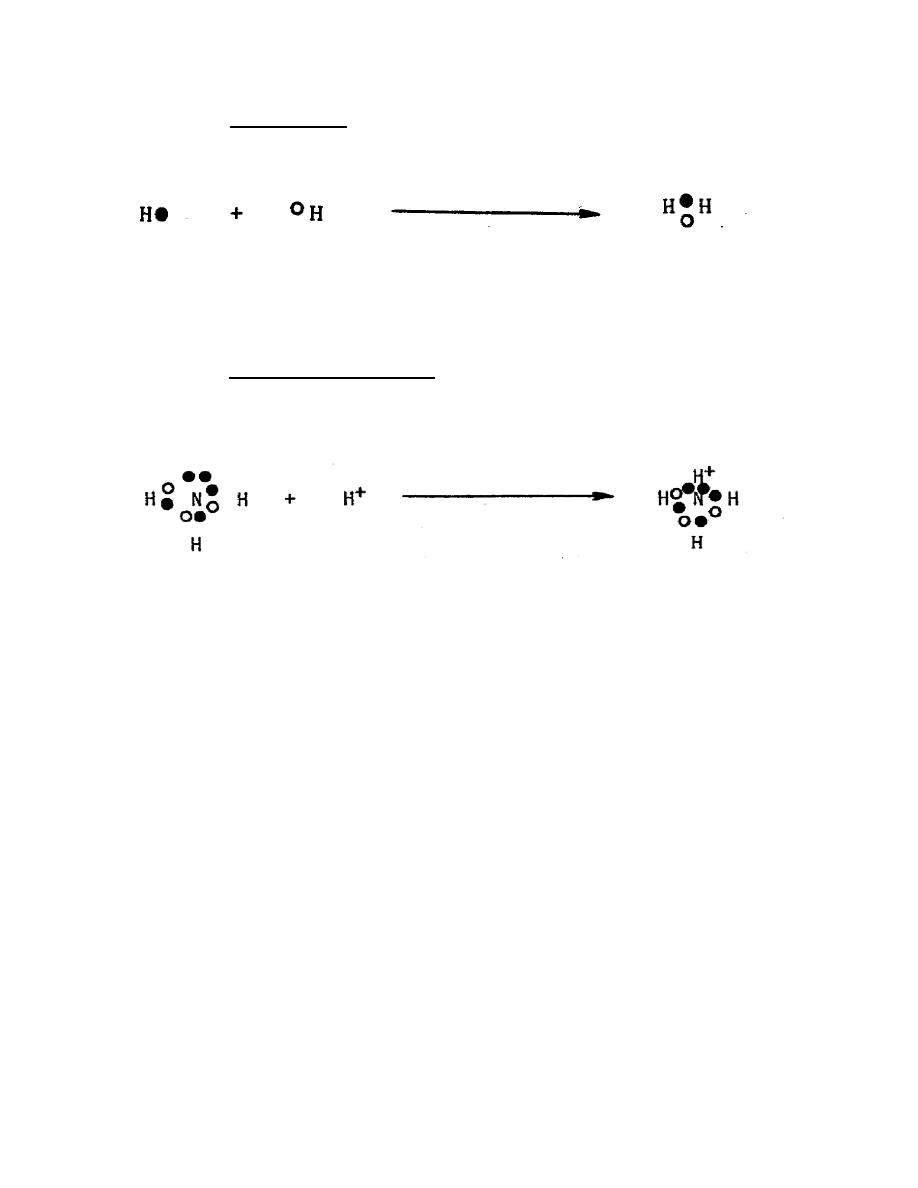
(2) Covalent bond. If two atoms each donate an electron that is shared with
the other atom, the bond is a covalent bond. An example of this is the bond between
two H (hydrogen) atoms. Double and triple covalent bonds are also possible.
Both atoms have 1 e- in the K shell.
By sharing their electrons each
-
hydrogen has 2 e in the K shell and
both are stable because of the
completed shell.
(3) Coordinate covalent bond. If one atom donates two electrons for sharing
with another atom (which donates no electrons), it is called a coordinate covalent bond.
(proton).
+
-
Ammonia -N
H has an
By sharing the two e ,
has a complete
empty K
H+ has a completed K shell
outer shell
shell
and N still has a completed
outer shell.
MD0803
1-16



 Previous Page
Previous Page
