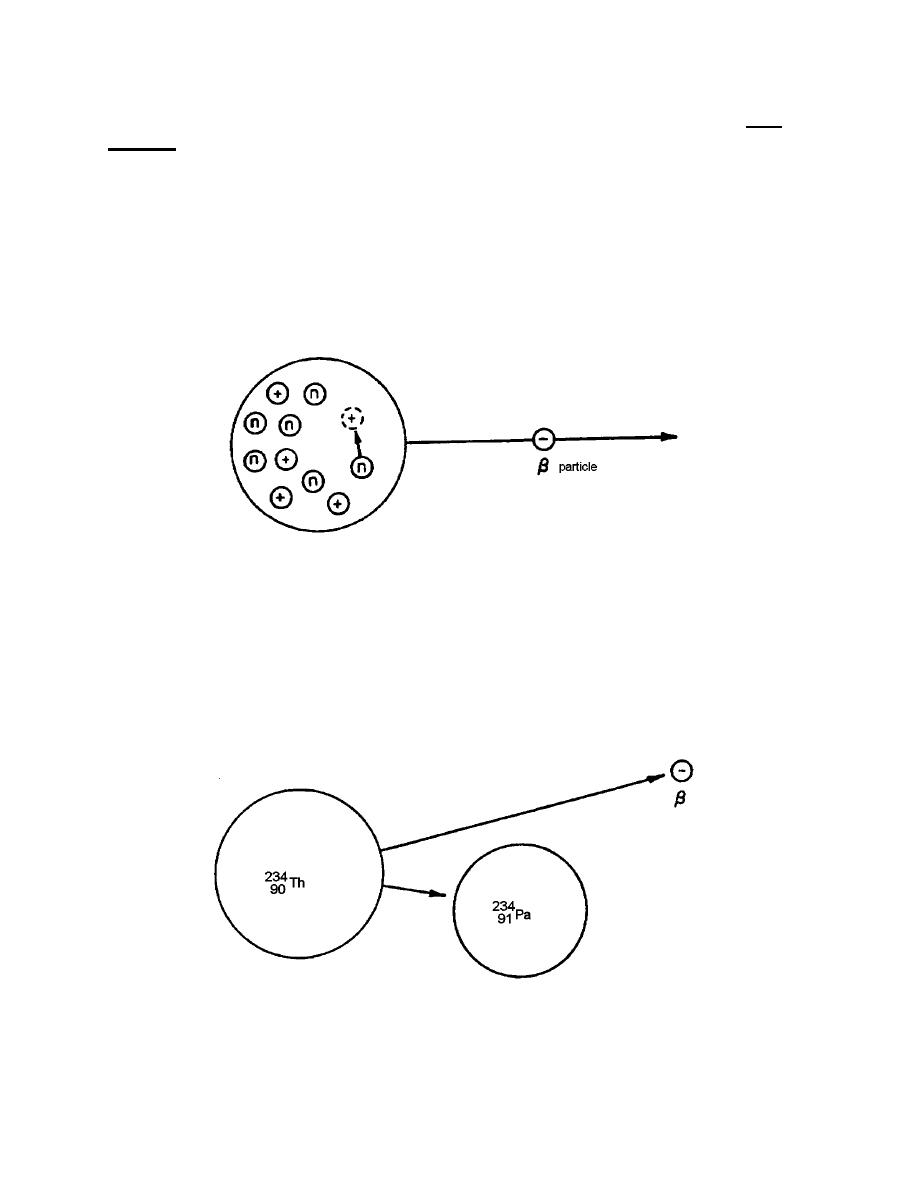
c. It often happens that unstable nuclei emit high-speed electrons called beta
particles (symbol β). They are called particles, not electrons, to indicate that they are
electrons that originate in the nucleus instead of electrons that originate in atomic orbits
outside the nucleus. The source of electrons in this form of radioactivity is interesting
since, as was previously pointed out, there are no electrons in a nucleus. This paradox
is explained when it is found that a neutron can transform into a proton and an electron.
When this happens, the electron (β particle) is ejected from the nucleus while the proton
is left behind. Figure 1-7 pictures a hypothetical example of emission showing the
decay of a neutron into a proton. The proton remains within the nucleus and an
electron escapes.
Figure 1-7. Beta emission.
d. The thorium isotope that was formed in the example on emission is also
radioactive, but it is an emitter causing Th-234 to change to the element protactinium
(Figure 1-8). Note that the protactinium mass number (A = 234) does not change in
decay, but that the atomic number Z does increase from 90 to 91 because a proton has
been gained. This gain comes about, as explained earlier, when the neutron breaks
down, ejecting an electron and retaining the proton.
Figure 1-8. Beta emission from Th-234.
MD0180
1-11



 Previous Page
Previous Page
