1-6. CHEMICAL PROPERTIES
a. The electron structure of an atom also determines the chemical properties of
an atom. So far we have talked only about filling up the K-shell. In order to fill up the L-
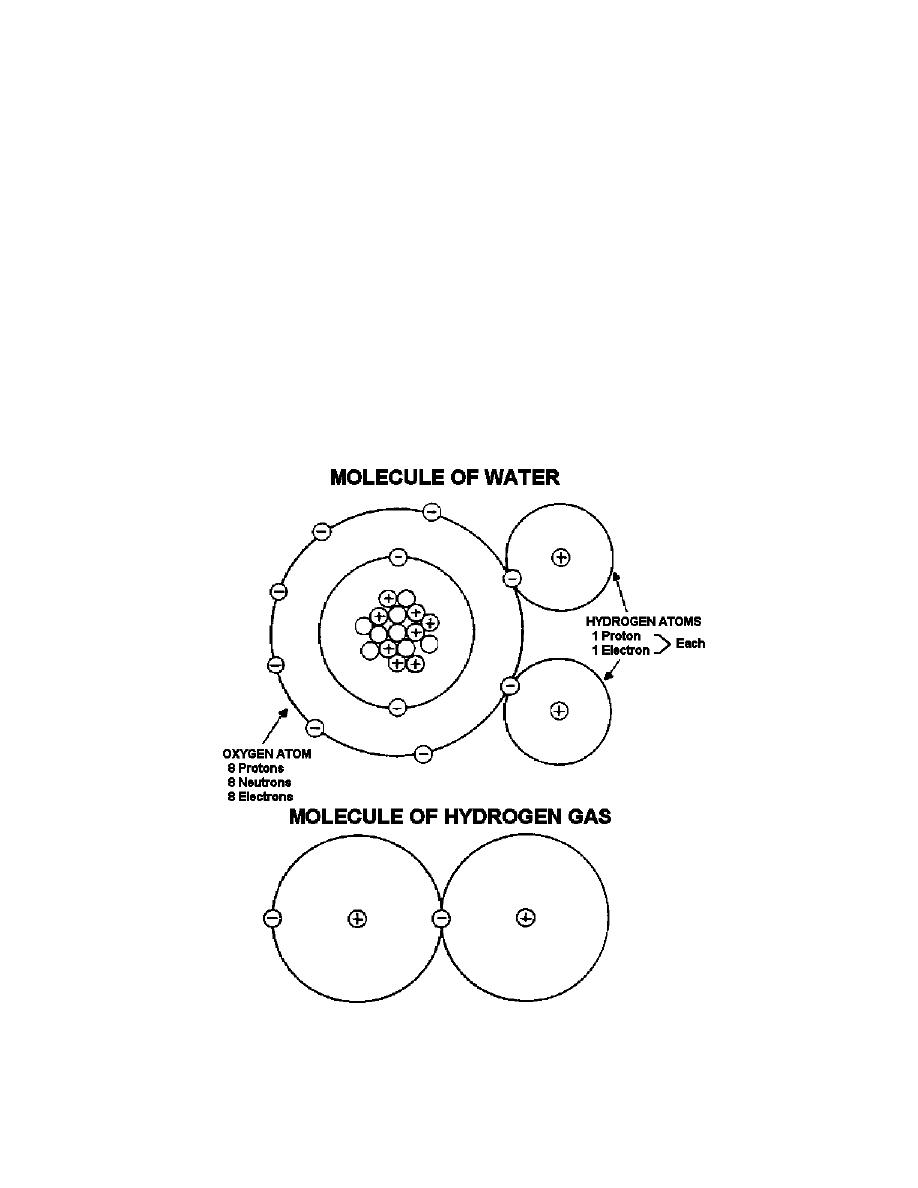
shell, eight electrons are required. Let us look at an oxygen atom; this has eight
protons, eight neutrons, and, therefore, eight electrons revolving about the nucleus.
Two of the eight electrons will be found in the K-shell. This means that six are in the
L-shell in a normal oxygen atom. In order to fill the L-shell and make it complete, two
more electrons would be needed. Now most atoms would like to fill up their outer shell;
but for oxygen, this would mean that it would have a total negative charge of -2. It is
possible that the oxygen atom could borrow enough electrons from hydrogen atoms to
complete the L-shell. Two hydrogen atoms would be needed. The oxygen atom would
have a net charge of 2 and each hydrogen atom would then be left with a positive, +1,
charge. Since positive attracts negative, this group of particles would tend to hold
together. This form of chemical combination is known as ionic bonding. This hydrogen-
oxygen group is called a molecule of water (see Figure 1-5).
Figure 1-5. Descriptive pictures of molecules.
MD0180
1-8



 Previous Page
Previous Page
