(acid) concentration in the blood (or in the body) and an arbitrary number. The
balancing part (base or alkaline) is a hydroxy group (OH ). The normal range of the
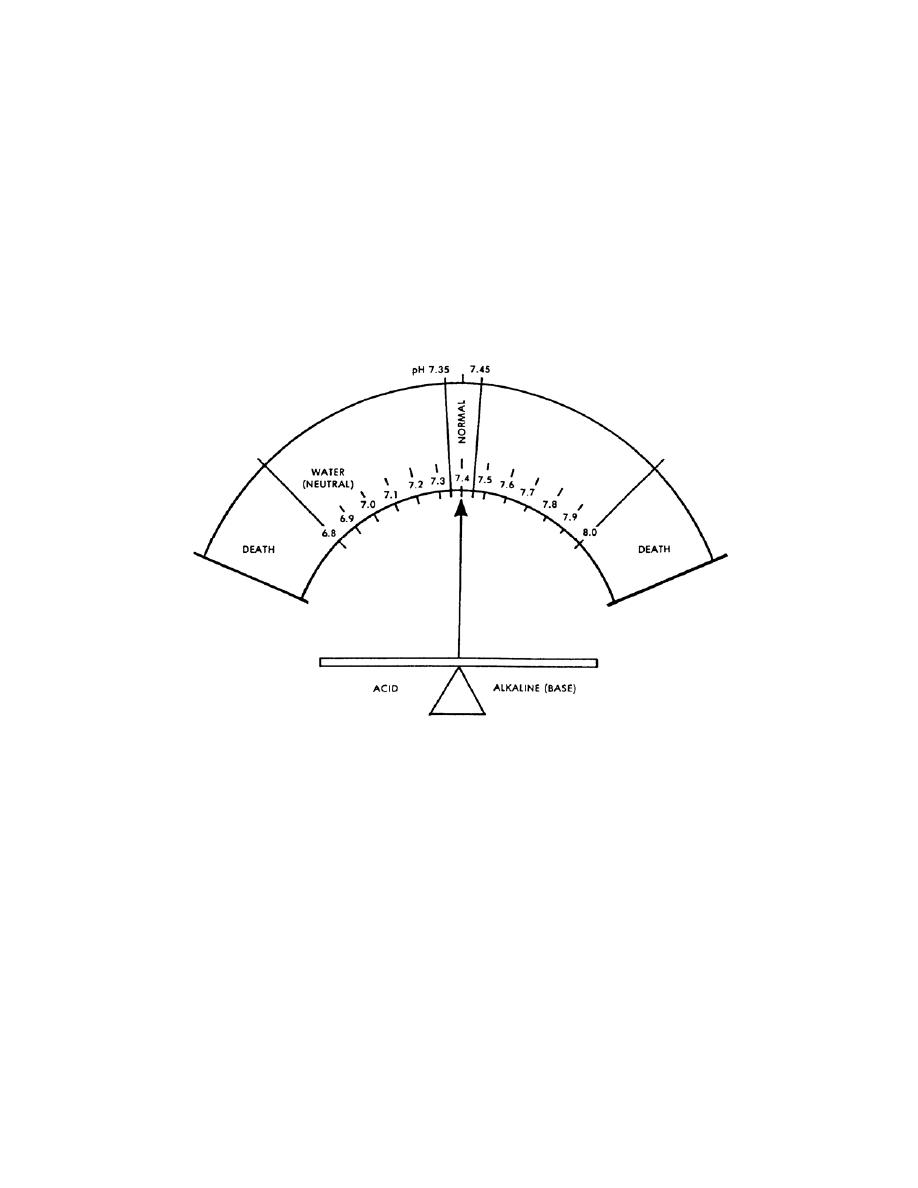
blood pH is 7.35 to 7.45. There is a daily variation caused by production of acids by
exercise and metabolism of food. In the terminal stages of some diseases, pH can vary
from a low of 6.8 to a high of 7.8. The concept of pH may be easier to understand by
comparing the acid (hydrogen) and base (hydroxl) factors to pure water, which is neutral
(neither acid nor base). The body is always slightly alkaline. The body's acid-base
balance is effectively maintained under normal circumstances by the various buffer
processes which neutralize strong acids or strong bases (alkalines) using the body's
various buffer systems (chemical, organic, and so forth) to help excrete excess body
system products. Figure 2-3 shows the narrow range of the body's pH.
Figure 2-3. Acid-base balance.
2-7.
NATURE OF SOLUTIONS
There are three movement directions possible following the introduction of
injectable solutions into the body. These movement directions are governed by the
nature of the solution with regard to body fluids. The fluids are called hypotonic,
hypertonic, and isotonic.
a. Hypotonic Solutions. A hypotonic solution is one that has less tonicity than
the fluid within the body's cells. This type of fluid is absorbed into the body's cells by
moving across the cell's membrane and into the cell. If too much hypotonic solution is
added, there is always the danger that the cells could burst or at least become irritated.
Examples of hypotonic solutions are 0.45 percent sodium chloride solution and sterile
water. This movement is shown in figure 2-4.
MD0564
2-7




 Previous Page
Previous Page
