1-12. ISOTOPES
a. Atoms of the same element (having the same atomic number) but having
different mass numbers are known as isotopes. Isotopes have the same number of
protons but a different number of neutrons in their nuclei. Consequently, the various
isotopes have different atomic weights. Because an element consists of a group of
isotopes, the atomic weight is an average and almost never a whole number.
b. For example, zinc with an atomic number of 30 has an atomic mass of 65.38,
definite proportions. The terms atomic weight and mass number are often used
interchangeably because they are nearly equal to each other.
c. The term isotope does not always refer to a distinct species of atom, but the
term nuclide does. It refers to a species of atom characterized by its nuclear
configuration, and hence by the number of protons, the number of neutrons, and the
energy content. We can say that the different isotopes of an element are composed of
nuclides that have the same atomic number but different mass numbers.
1-13. VALENCE
a. The number of electrons in the outer shell of an atom determines its valence.
Valence represents the capacity of an atom to combine with other atoms to form
molecules. The hydrogen atom with a valence of +1 is the standard used for
determining the valence of other atoms. Atoms with the same number of outer orbital
electrons have similar chemical and physical characteristics.
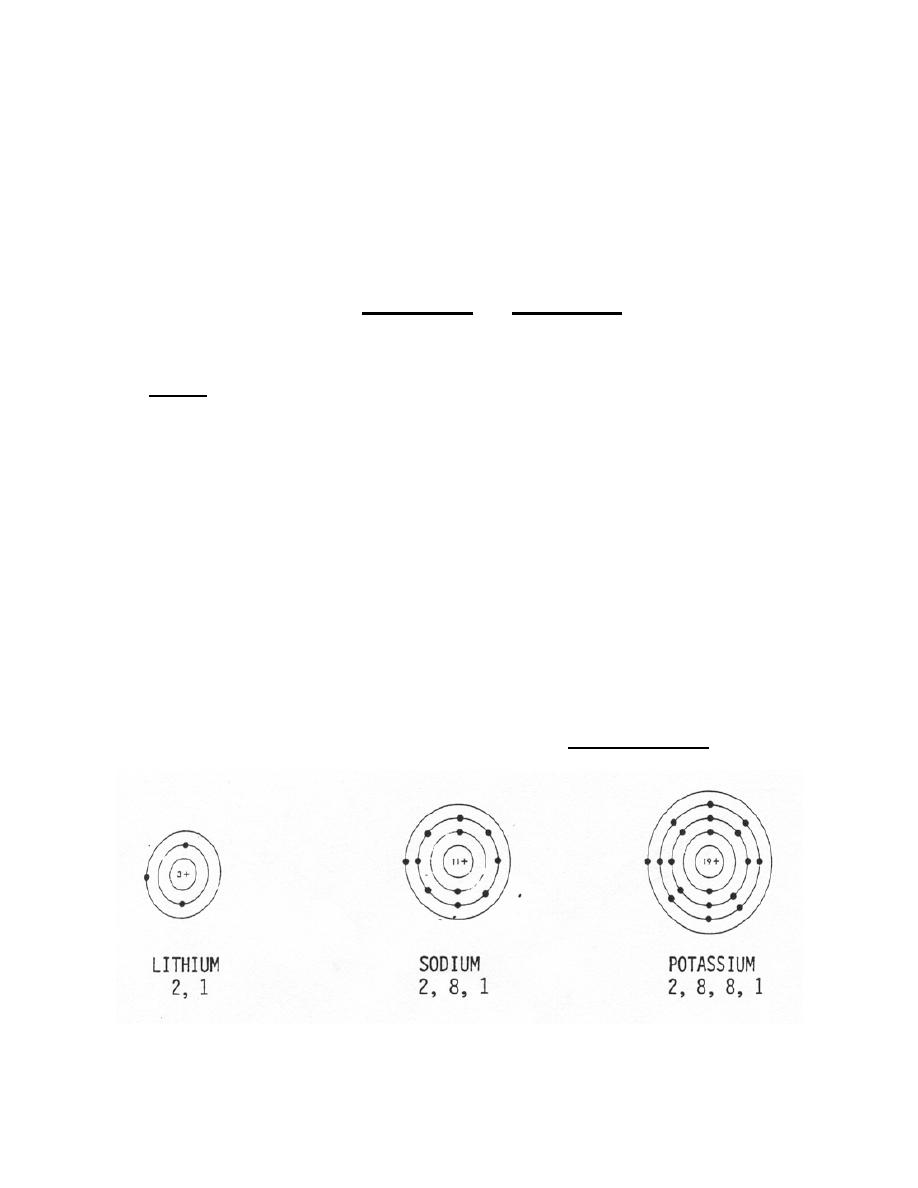
b. For example, atoms of elements such as lithium, sodium, and potassium have
only one electron in their outermost shells (figure 1-5). Their valence is +1, and they
combine with other elements that may lack a single electron in the outermost shell.
Atoms lacking one electron to complete their outermost orbit are said to have a valence
of -1 (figure 1-6). These give-or-take electrons are called valence electrons.
Figure 1-5. Atoms with a +1 valence.
MD0950
1-12



 Previous Page
Previous Page
