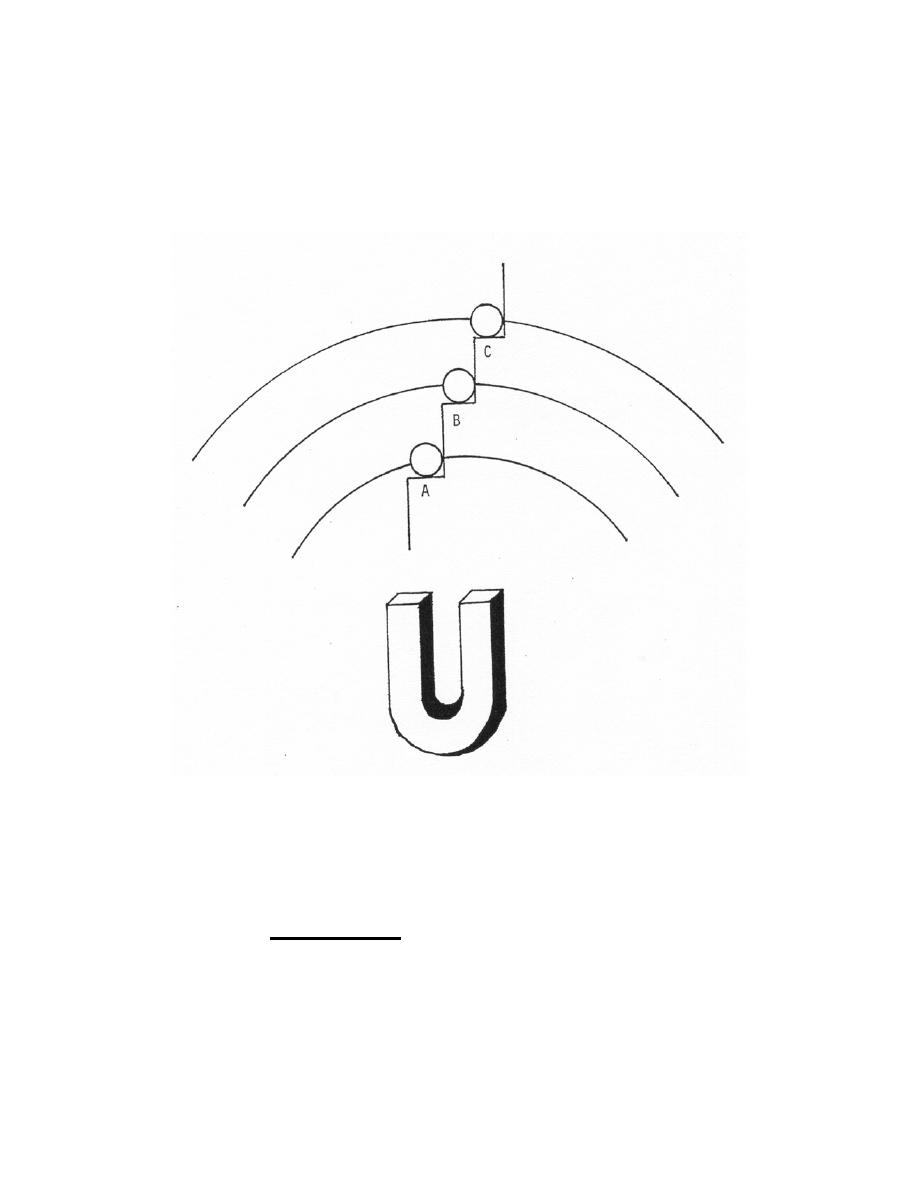
from the magnet as illustrated in figure 1-4. The magnetic force between the magnet
and ball would be similar to the force of attraction between the nucleus and an electron
since the force is inversely proportional to the square of the distance between the two.
Thus, the ball on step A would be more firmly held (as a K shell electron) than the ball in
step B (as an L shell electron) and, correspondingly, different amounts of energy would
be required to remove them.
Figure 1-4. Binding and potential energy.
Binding and potential energy varies with object distance from a magnetic force.
Thus, the distance between orbiting electrons and the atomic nucleus affects
both the binding energy and the potential energy.
(b) Potential energy. The potential energy of the electrons is similar to
the potential energy of the metal balls (figure 1-4). The ball on step C has more
potential energy than the ball on step B because its position is more elevated. If the ball
on Step C dropped to step B, it would release a certain amount of energy. Orbiting
electrons also release energy in the same manner when they drop from a higher
potential energy level to a lower potential energy level. Thus, the potential energy is
higher when there are more electrons in shells farther from the nucleus.
MD0950
1-11



 Previous Page
Previous Page
