Figure 1-6. Atoms with 1 valence.
1-14. FORMATION OF MOLECULES
Molecules are formed of two or more atoms. Sharing of the outer valence
electrons often occurs among atoms during molecule formation. This sharing is
cemented by three kinds of bonds: the ionic or chemical bond, the convalent bond, and
he hydrogen.
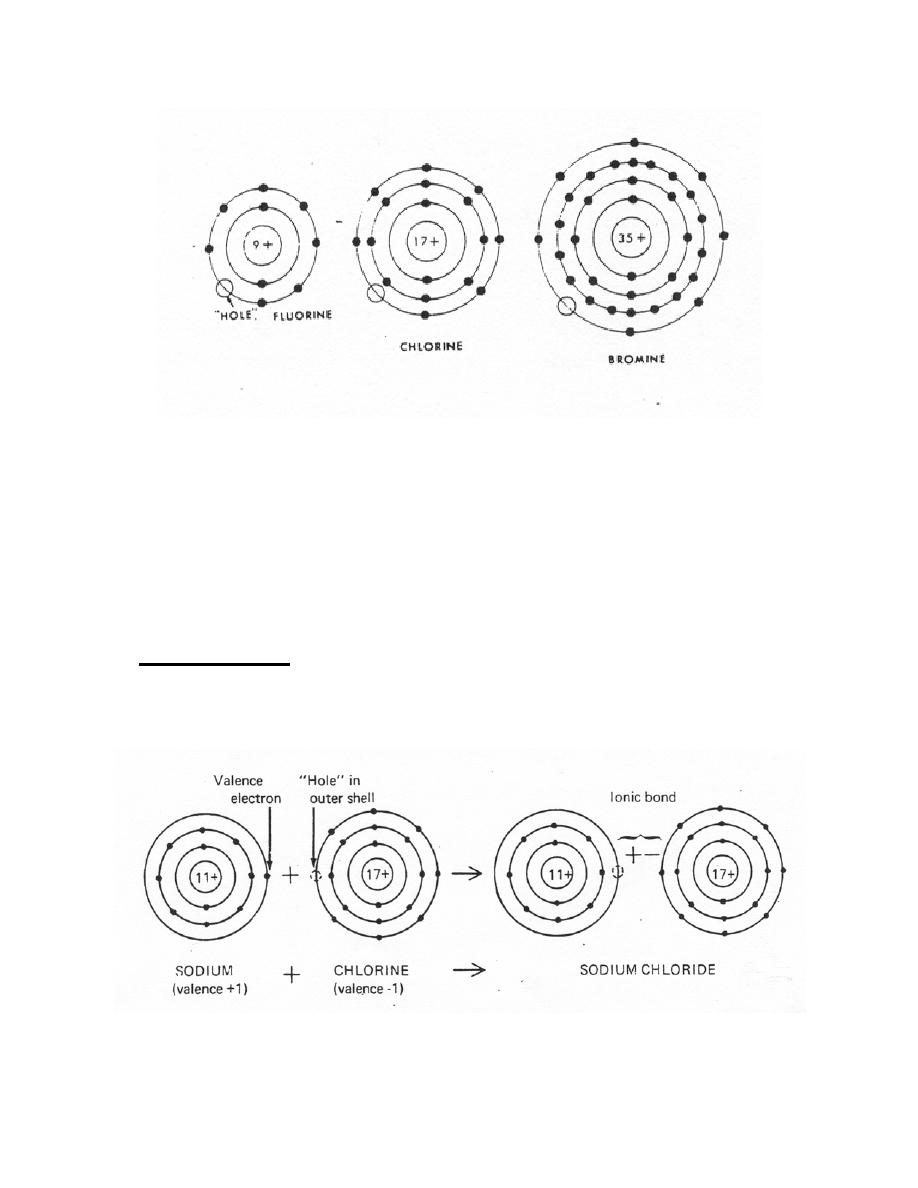
a. Ionic Bond. An ionic bond is formed with the direct transfer of an orbital
electron from one atom to another in an electrostatic attraction. This type of valence,
call ionic polar valence, is common to the formation of many inorganic compounds. For
example, when a sodium atom and a chlorine atom are brought together, the single-
valence electron in the outer shell of the sodium atom is transferred to the chlorine atom
which has a "hole" in its outer shell and sodium chloride is formed (figure 1-7).
Figure 1-7. Ionic bond sodium chloride.
MD0950
1-13



 Previous Page
Previous Page
