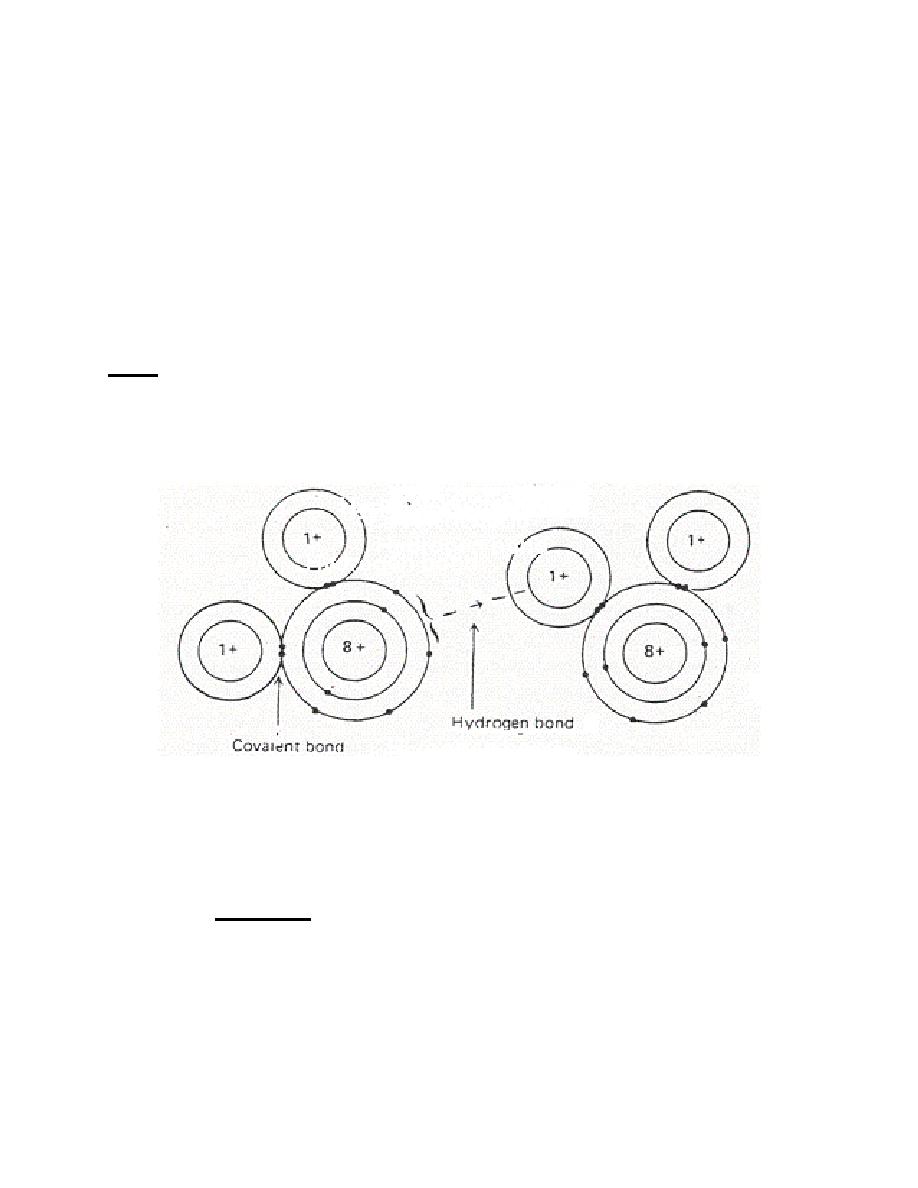
c. Hydrogen Bond. A hydrogen bond is an attraction between molecules that
is weaker than ionic or covalent bonds.
(1) This may be illustrated by the intermolecular attraction between
angular placement of the hydrogen atoms on the oxygen atom sets up a polar
establishes geometric arrangements and is an electrostatic phenomenon.
(2) Long chains of protein molecules containing nitrogen and oxygen atoms
are also held together by hydrogen bonds.
NOTE:
It will be noticed by now that the chemical reactions in molecule formation
follow a pattern of definite proportions that may be explained by the concept
of valence. Examples: sodium chloride is always formed by combining one
atom of sodium with one atom of chlorine; water always by two atoms of
hydrogen with one atom of oxygen.
Figure 1-10. Hydrogen bond between two molecules of water.
1-15. RADIOACTIVITY
The spontaneous disintegration of radioactive substances in which the atomic
nuclei undergo partial breakdown and give off penetrating radiation at the same time is
described as radioactivity. It is a natural property of all existing elements with atomic
numbers above 83. It is also possible to induce this property in all other known
elements.
a. A factor contributing to nuclear disintegration in radioelements is the instability
of their atomic nuclei. For these elements to reach a more stable or less energetic
state, excess energy in the form of alpha, beta, or gamma radiation is released. This
nuclear instability may occur from the natural configuration of the atoms of the element
or it may be man-made.
MD0950
1-15



 Previous Page
Previous Page
